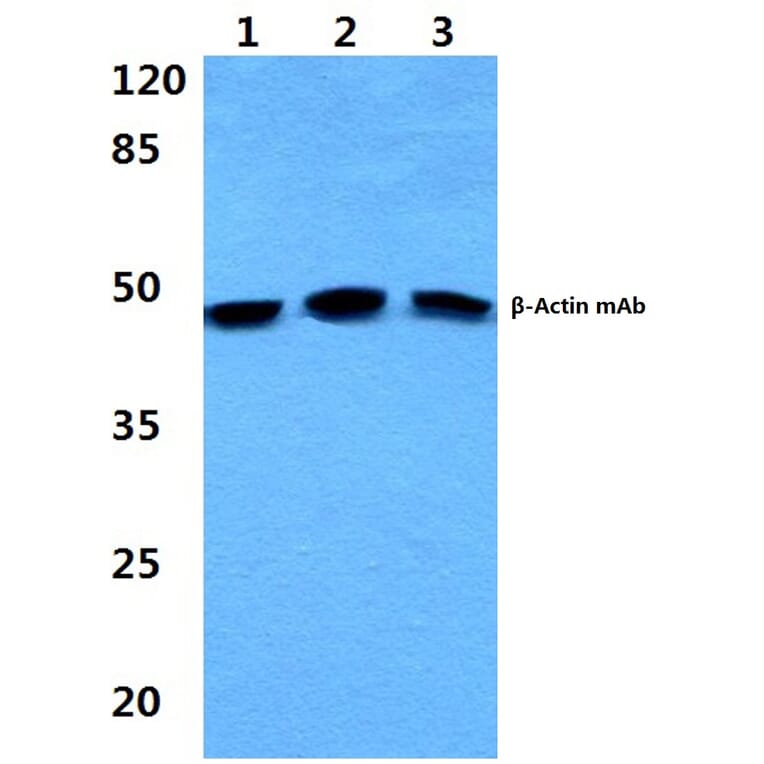
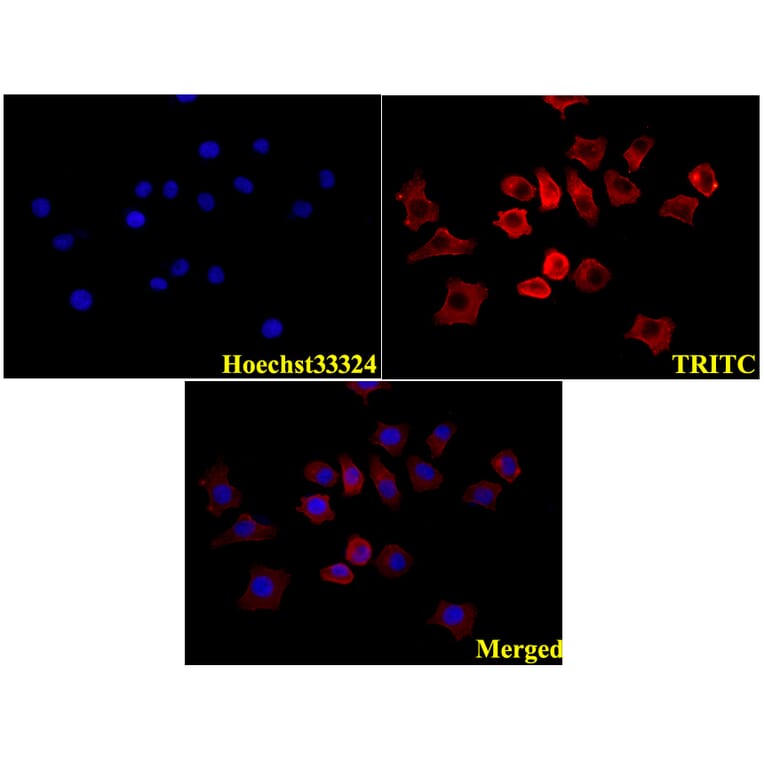
Unconjugated
The NF-E2-related factor 2 (Nrf2)/antioxidant response element (ARE) pathway plays a critical role in protecting against oxidative stress in brain ischemia and reperfusion injury. Glycogen synthase kinase 3β (GSK-3β) may play a critical role in regulating Nrf2 in a Kelch-like ECH-associated protein 1 (Keap1)-independent manner. However, the relationship between GSK-3β and Nrf2 in brain ischemia and reperfusion injury is not clear. In this study, we explored the mechanisms through which GSK-3β regulates Nrf2 and Nrf-2/ARE pathways in vitro and in vivo. We used oxygen and glucose deprivation/reoxygenation (OGD/R) in primary cultured cortical neurons and a middle cerebral artery occlusion-reperfusion (MCAO/R) rat model to mimic ischemic insult. In this study, GSK-3β siRNA and inhibitors (SB216763 and LiCl) were used to inhibit GSK-3β in vitro and in vivo. After inhibiting GSK-3β, expression of total and nuclear Nrf2, Nrf2-ARE binding activity, and expression of Nrf2/ARE pathway-driven genes HO-1 and NQO-1 increased. Overexpression of GSK-3β yielded opposite results. These results suggest that GSK-3β downregulates Nrf2 and the Nrf2/ARE pathway in brain ischemia and reperfusion injury. GSK-3β may be an endogenous antioxidant relevant protein, and may represent a new therapeutic target in treatment of ischemia and reperfusion injury.
The rabies virus (RABV) glycoprotein (G) is the principal antigen responsible for the induction of virus neutralizing antibodies (VNA) and is the major modality of protective immunity in animals. A recombinant RABV HEP-Flury strain was generated by reverse genetics to encode two copies of the G-gene (referred to as HEP-dG). The biological properties of HEP-dG were compared to those of the parental virus (HEP-Flury strain). The HEP-dG recombinant virus grew 100 times more efficiently in BHK-21 cell than the parental virus, yet the virulence of the dG recombinant virus in suckling mice was lower than the parental virus. The HEP-dG virus can improve the expression of G-gene mRNA and the G protein and produce more offspring viruses in cells. The amount of G protein revealed a positive relationship with immunogenicity in mice and dogs. The inactivated HEP-dG recombinant virus induced higher levels of VNA and conferred better protection against virulent RABV in mice and dogs than the inactivated parental virus and a commercial vaccine. The protective antibody persisted for at least 12 months. These data demonstrate that the HEP-dG is stable, induces a strong VNA response and confers protective immunity more effectively than the RABV HEP-Flury strain. HEP-dG could be a potential candidate in the development of novel inactivated rabies vaccines.
Osterix (Osx/SP7) is a C2H2 zinc finger-containing transcription factor of the SP gene family. Osx knockout mice indicate that the gene plays an essential role in osteoblast differentiation and bone formation. However, the mechanisms involved in the regulation of Osx are still poorly understood. Here, we report a novel post-translational mechanism for the regulation of Osx in mammalian cells. We found that the stability of endogenous and exogenous Osx reduced after cycloheximide treatment. In cells treated with the proteasome inhibitors MG-132 or lactacystin, both endogenous and exogenous Osx protein expression increased in a time-dependent manner. Co-immunoprecipitation (Co-IP) assays showed that both endogenous and exogenous Osx were ubiquitinated. Six lysine residues of Osx were identified as candidate ubiquitination sites by construction of point mutant plasmids and luciferase reporter assays. Furthermore, we confirmed that K58 and K230 are the ubiquitination sites of Osx by Co-IP assays and protein stability assays. Moreover, the Osx K58R and K230R mutations promoted the expression of osteoblast differentiation markers (alkaline phosphatase, collagen I and osteocalcin) and enhanced osteogenic differentiation in C2C12 cells. Taken together, our data indicate that Osx is an unstable protein, and that the ubiquitin-proteasome pathway is involved in the regulation of Osx and thereby regulates osteoblast differentiation.
BACKGROUND:
Glioblastoma is an extraordinarily aggressive disease that requires more effective therapeutic options. Snail family zinc finger 1, dysregulated in many neoplasms, has been reported to be involved in gliomas. However, the biological mechanisms underlying SNAI1 function in gliomas need further investigation.
METHODS:
Quantitative real-time PCR was used to measure microRNA-128 (miR-128) expression level and western blot was performed to detect protein expression in U87 and U251 cells and human brain tissues. Cell cycle, CCK-8, transwell and wound-healing assays were performed. Dual-luciferase reporter assay was used for identifying the mechanism of SNAI1 and miR-128b regulation. The mechanism of miR-128 targeting SP1 was also tested by luciferase reporter assay. Immunohistochemistry and in situ hybridisation staining were used for quantifying SNAI1, SP1 and miR-128 expression levels in human glioma samples.
RESULTS:
The Chinese Glioma Genome Atlas (CGGA) data revealed that SNAI1 was up-regulated in glioma and we confirmed the findings in normal and glioma tissues. SNAI1 depletion by shRNA retarded the cell cycle and suppressed proliferation and invasion in glioma cell lines. The CGGA data showed that the Pearson correlation index between SNAI1 and miR-128 was negatively correlated. SNAI1 suppressed miR-128b expression by binding to the miR-128b specific promoter motif, and miR-128 targeted SP1 via binding to the 3'-untranslated region of SP1. Moreover, introduction of miR-128 anti-sense oligonucleotide alleviated the cell cycle retardation, proliferation and invasion inhibition induced by SNAI1 shRNA. Immunohistochemistry and in situ hybridisation analysis of SNAI1, SP1 and miR-128 unraveled their expression levels and correlations in glioma samples.
CONCLUSIONS:
We propose that the SNAI1/miR-128/SP1 axis, which plays a vital role in glioma progression, may come to be a clinically relevant therapeutic target.
The present study aimed to investigate bone microarchitecture of the proximal tibia in glucocorticoid-induced osteoporosis (GIOP) mice, and the underlying molecular mechanisms of curcumin in DXM-induced osteoporosis were performed. DXM-treated facilitated to induce hypercalciuria in mice, and curcumin-treated showed a decrease in urine calcium. Curcumin reversed DXM-induced bone resorption, including an increase in serum OCN and a decrease in bone resorption markers CTX and TRAP-5b. H&E staining showed the increased disconnections and separation in trabecular bone network as well as the reduction of trabecular thickness throughout the proximal metaphysis of tibia in GIOP group. Importantly, curcumin reversed DXM-induced trabecular deleterious effects and stimulated bone remodeling. The further evidence showed that curcumin supplement significantly decreased the TRAP-positive stained area and inhibited the activity of OPG/RANKL/RANK signaling in the GIOP mice. Moreover, bioinformatics analysis suggested that miR-365 was a regulator of MMP9. The levels of miR-365 were markedly suppressed; however, curcumin treatment could reverse the downregulation of miR-365 in the tibia of GIOP mice. Simultaneously, the results demonstrated that the mRNA and protein expression of MMP-9 were significantly increased in GIOP mice compared with that of the control group. Curcumin treatment could suppress the expression of MMP-9 in the tibia of GIOP mice. The present study demonstrated the protective effects of curcumin against bone deteriorations in the experimentally DIOP mice, and the underlying mechanism was mediated, at least partially, through the activation of microRNA-365 via suppressing MMP9.
Enterovirus 71 (EV71), the etiological agent of hand-foot-and-mouth disease, has increasingly become a public health challenge around the world. Previous studies reported that EV71 infection can induce autophagic machinery to enhance viral replication in vitro and in vivo, but did not address the underlying mechanisms. Increasing evidence suggests that autophagy, in a virus-specific manner, may function to degrade viruses or facilitate viral replication. In this study, we reported that EV71 infection of human epidermoid carcinoma (Hep2) and African green monkey kidney cells (Vero) induced autophagy, which is beneficial for viral replication. Our investigation of the mechanisms revealed that EV71 infection resulted in the reduction of cellular miR-30a, which led to the inhibition of Beclin-1, a key autophagy-promoting gene that plays important roles at the early phase of autophagosome formation. We provided further evidence that by modulating cellular miR-30a level through either overexpression or inhibition, one can inhibit or promote EV71 replication, respectively, through regulating autophagic activity.
Acetylcholinesterase (AChE) is impaired in brain of diabetic animals, which may be one of the reasons for diabetes-associated cognitive decline. However, the mechanism is still unknown. The present study was designed to investigate whether the increased expression of AChE in central neurons under high glucose (HG) condition was due to activation of mammalian target of rapamycin (mTOR) signaling. It was found that more production of reactive oxygen species, and higher levels of phospho-Akt, phospho-mTOR, phospho-p70S6K, and AChE were detected in HT-22 cells in HG group than normal glucose group after culture for 24 h, which were all attenuated by an antioxidant N-acetyl-L-cysteine. A PI3K inhibitor LY294002 significantly decreased the levels of phospho-Akt, phospho-mTOR, phospho-p70S6K, and AChE protein expression in HG-cultured HT-22 cells, and an mTOR inhibitor rapamycin markedly reduced the levels of phospho-mTOR, phospho-p70S6K, and AChE expression. Furthermore, compared with normal rats, diabetic rats showed remarkable increases in levels of AChE activity and expression, malondialdehyde, phospho-mTOR, phospho-p70S6K, and a significant decrease in total superoxide dismutase activity in both hippocampus and cerebral cortex. However, much lower levels of phospho-mTOR, phospho-p70S6K, and AChE expression occurred in both brain regions of diabetic rats treated with rapamycin when compared with untreated ones. These results indicated that mTOR signaling was activated through the activation of PI3K/Akt pathway mediated by oxidative stress in HG-cultured HT-22 cells and diabetic rat brains, which contributed to the elevated protein expression of AChE in central neurons under the condition of HG.
Geniposide (GP), an iridoid glucoside extracted from Gardenia jasminoides Ellis fruits, has been used as a herbal medicine to treat liver and gall bladder disorders for many years. However the mechanism of anti-inflammatory is largely unknown. In this study, GP significantly attenuated inflammation in acute liver injury (ALI) mice model and in lipopolysaccharide (LPS)-induced THP-1 cells. It was demonstrated that GP obviously decreased the expression of Methyl-CpG binding protein 2 (MeCP2) in vivo and in vitro. Knockdown of MeCP2 with siRNA suppressed the expressions of IL-6 and TNF-α, while over-expression of MeCP2 had a proinflammatory effect on the expression of IL-6 and TNF-α in LPS-induced THP-1 cells. Mechanistically, it was indicated that GP had anti-inflammatory effects at least in part, through suppressing MeCP2. Interestingly, GP could attenuate expressions of Sonic hedgehog (Shh) and GLIS family zinc finger 1 (GLIS1) but increase Ptched1 (PTCH1) expression. Similar findings were also demonstrated at the protein level by siRNA MeCP2. Furthermore, over-expression of MeCP2 obviously increased Shh and GLIS1 expressions but reduced PTCH1 expression. Taken together, GP may serve as an effective modulator of MeCP2-hedgehog pathway (Hh)-axis during the pathogenesis of inflammation. Our findings shed light on the potential therapeutic feature of GP in recovering inflammatory diseases.
Toll-like receptor-4 (TLR4) signaling has been implicated in innate immunity and acute inflammation following acute lung injury (ALI). As such, modulating inflammatory response through TLR4 represents an attractive therapeutic approach to treat ALI. Increasing evidence demonstrates that hyaluronan (HA) can modulate TLR4 activation and has shown early promise as a therapeutic agent in ALI. However, the mechanism associated with HA has not been fully elucidated. In the current study, we sought to determine the effects of HA on lipopolysaccharide (LPS)-induced inflammatory response and gain insights into the mechanism of action in mice with intratracheal instillation of LPS. Our results demonstrate that in contrast to mice challenged with LPS, pretreatment with HA significantly inhibited inflammatory cell recruitment, attenuated lung injury and suppressed the level of cytokine/chemokine in bronchial alveolar lavage fluid (BALF). Investigation of the mechanism responsible for inhibition of LPS activation showed HA treatment significantly inhibited the nuclear translocation of NF-κB p65 and protein expression of myeloid differentiation primary response protein (MyD88) and TIR-domain-containing adapter-inducing interferon-β (TRIF) and p38 MAPK, JNK and ERK activation in lung tissue. Furthermore, we compared the protection effect of HA in TLR4-deficient mice with those of genetically matched wild type (WT) mice in an acute model of lung injury. However, in TLR4-deficient mice, HA pretreatment before LPS instillation fail to affect the LPS response. Therefore, our findings suggest that HA pretreatment attenuated LPS-induced ALI and the anti-inflammatory function of HA was partial dependent on TLR4, which shed new light on potential elements that regulate the lung injury response.
The human lens is comprised largely of crystallin proteins assembled into a highly ordered, interactive macro-structure essential for lens transparency and refractive index. Any disruption of intra- or inter-protein interactions will alter this delicate structure, exposing hydrophobic surfaces, with consequent protein aggregation and cataract formation. Cataracts are the most common cause of blindness worldwide, affecting tens of millions of people, and currently the only treatment is surgical removal of cataractous lenses. The precise mechanisms by which lens proteins both prevent aggregation and maintain lens transparency are largely unknown. Lanosterol is an amphipathic molecule enriched in the lens. It is synthesized by lanosterol synthase (LSS) in a key cyclization reaction of a cholesterol synthesis pathway. Here we identify two distinct homozygous LSS missense mutations (W581R and G588S) in two families with extensive congenital cataracts. Both of these mutations affect highly conserved amino acid residues and impair key catalytic functions of LSS. Engineered expression of wild-type, but not mutant, LSS prevents intracellular protein aggregation of various cataract-causing mutant crystallins. Treatment by lanosterol, but not cholesterol, significantly decreased preformed protein aggregates both in vitro and in cell-transfection experiments. We further show that lanosterol treatment could reduce cataract severity and increase transparency in dissected rabbit cataractous lenses in vitro and cataract severity in vivo in dogs. Our study identifies lanosterol as a key molecule in the prevention of lens protein aggregation and points to a novel strategy for cataract prevention and treatment.
Hepatocellular carcinoma (HCC) is the fifth most common cancer in men and the seventh in women worldwide. Chronic heavy alcohol consumption is a major risk factor for the development of HCC. However, the mechanism underlying the direct association between alcohol consumption and HCC is far from completely understood. In the present study, we investigated the effect of chronic consumption of alcohol on diethylnitrosamine (DEN)-induced cytotoxicity, which was essential for the malignant transformation. We showed that alcohol deceased survival of mice treated by DEN and promoted DEN-induced toxicity and hepatic injury. In addition, alcohol promoted DEN-induced increase of proinflammatory factors, collagen content and fibrosis-related genes, including collagen1, 3 and 4, TMIP1, TIMP2 and TGFβ1, and compensatory proliferation. Alcohol may increase alcohol dehydrogenase (ADH) and cytochrome P4502E1 (CYP2E1) expression, enhanced reactive oxygen species (ROS) generation, and resulted in a vicious circle between ROS generation, lipid accumulation, and mitochondrial dysfunction, aggravating liver injury and toxicity in DEN-treated mice. These results demonstrated that the combination of alcohol and carcinogens could aggravate carcinogen-induced cytotoxicity in the early phase of rumourigenesis through ADH and CYP2E1-generated ROS and the resultant cytotoxic process. The present study provided direct experimental evidence for alcohol-promoted toxicity and hepatic injury in carcinogen (DEN)-treated mice.
Nowadays, people pay more and more attention to the natural products based on their multiple targets in the antitumor treatment. In our previous research, Rhizoma paridis saponins (RPS) were regarded as potent anticancer agent that elicits programmed cell death and inhibits metastases in murine lung adenocarcinoma in vivo. In the present study, we set up a rat model of diethylnitrosamine (DEN) induced pulmonary adenoma to evaluate the antitumor effects of RPS again. After 20 weeks treatment, rats were sacrificed in order to perform histopathological examinations, blood biochemistry, immunohistochemistry, western blot, PCR and metabonomics. As a result, DEN induced pulmonary adenoma generation in the lungs and damaged hepatocytes and hepatoma formation in the livers. RPS effectively attenuated hepatotoxic and inhibited pulmonary adenoma through down-regulating expression of MMP-9 and up-regulating level of TIMP-2 in DEN-induced rats. Meanwhile, RPS remarkably decreased energy metabolism, and glycine, serine and threonine metabolism to block the tumor growth. In conclusion, RPS would be a potent anticancer agent used in the prospective application.
Hepatocellular carcinoma (HCC) has a high mortality rate worldwide and still remains to be a noticeable public health problem. Therefore, new remedies are urgently needed. Melittin, a major component of bee venom, is known to suppress cell growth in various cancers including HCC. However, the mechanism of the anticancer effect of melittin on HCC has not been fully elucidated. It has been reported that Methyl-CpG binding protein 2 (MeCP2) plays a key role in tumor proliferation, apoptosis, migration and invasion. In the present study, we found the high expression of MeCP2 in human HCC tissues and in the SMMC-7721 cell line. MeCP2 silencing inhibited cell proliferation, while over-expression of MeCP2 promoted cell growth in SMMC-7721 cells. It indicates that MeCP2 may be an attractive target for human HCC. We further found that melittin could inhibit cell proliferation by reducing MeCP2 expression in vitro. Interestingly, the inhibitory effect of melittin on cell proliferation was due to a delay in G0/G1 cell cycle progression, without influencing cell apoptosis. Next, we investigated the potential molecular mechanisms and found that MeCP2 could modulate Shh signaling in SMMC-7721 cells. Further study indicates that melittin may induce the demethylation of PTCH1 promoter, resulting in the increased expression of PTCH1. Furthermore, the expression of Shh and GLI1 was significantly lowered upon treatment of melittin. These results suggest that melittin can block Shh signaling in vitro. In short, these results indicate that melittin inhibits cell proliferation by down-regulating MeCP2 through Shh signaling in SMMC-7721 cells.
L-Theanine is an amino acid derivative from green tea. The present work was aimed at the effect of L-theanine on neuron-like rat pheochromocytoma (PC12) cells stimulated with cadmium chloride. Treatment with L-theanine before cadmium exposure increased cell viability; the experiments of Annexin V/PI staining indicated that L-theanine inhibited cadmium-induced cell apoptosis. Meanwhile, L-theanine decreased ROS production and protected from cadmium-induced disruption of mitochondrial transmembrane potential. Compared with cadmium-treated cells, L-theanine could also decrease the ratio of Bax/Bcl-2, as well as the level of cleaved caspase-9, caspase-3 and poly(ADP-ribose) polymerase. Furthermore, L-theanine depresses cadmium-induced up regulation of phosphorylations of PI3K/Akt, MAPK ERK1/2, and JNK signaling. These data suggest that L-theanine pretreatment reduces severity of cadmium toxicity probably via antioxidant action. Therefore, it may be concluded that L-theanine could be exploited for prevention of cadmium-induced diseases.
8-Chloro-cyclic AMP (8-Cl-cAMP) is a cyclic AMP analog that induces growth inhibition and apoptosis in a broad spectrum of cancer cells. Previously, we found that 8-Cl-cAMP-induced growth inhibition is mediated by AMP-activated protein kinase (AMPK) as well as p38 mitogen-activated protein kinase (p38 MAPK). To identify downstream mediators of the 8-Cl-cAMP signaling, we performed co-immunoprecipitation combined with mass spectrometry using the anti-AMPK or p38 MAPK antibodies. Through this approach, SHC1 was identified as one of the binding partners of p38 MAPK. SHC1 phosphorylation was suppressed by 8-Cl-cAMP in HeLa and MCF7 cancer cells, which was mediated by its metabolites, 8-Cl-adenosine and 8-Cl-ATP; however, 8-Cl-cAMP showed no effect on SHC1 phosphorylation in normal human fibroblasts. SHC1 siRNA induced AMPK and p38 MAPK phosphorylation and growth inhibition in cancer cells, and SHC1 overexpression re-sensitized human foreskin fibroblasts to the 8-Cl-cAMP treatment. SHC1 phosphorylation was unaffected by Compound C (an AMPK inhibitor) and SB203580 (a p38 MAPK inhibitor), which suggests that SHC1 is upstream of AMPK and p38 MAPK in the 8-Cl-cAMP-stimulated signaling cascade. On the basis of these findings, we conclude that SHC1 functions as a sensor during the 8-Cl-cAMP-induced growth inhibition in SHC1-overexpressing cancer cells.
Osteosarcoma (OS), the most common primary malignant bone tumor in children and adolescents, lacks an effective therapy. Stromal cell-derived factor (SDF-1) and its receptor, CXCR4, play multiple roles in migration, proliferation, and survival of different tumor cells. This study aimed to investigate whether the functional SDF-1/CXCR4 signaling mediates chemotaxis in F5M2 OS cells as well as the underlying mechanisms. Immunohistochemistry and immunofluorescence microscopy were used. RNA expression was detected by real-time quantitative polymerase chain reaction, and protein expression was examined by Western blotting. Migration assays were carried out in F5M2 cells. The results showed that the expression of CXCR4 and β-catenin mRNA and protein was significantly higher in OS tissues compared to the surrounding non-neoplastic tissues. SDF-1 promoted F5M2 cell migration by activating the AKT and Wnt/β-catenin signaling pathway, which was abrogated by preincubation with AMD3100 and LY294002. In conclusion, SDF-1/CXCR4 axis-promoted F5M2 cell migration was regulated by the Wnt/β-catenin signaling pathway.
Transforming growth factor-β1 (TGF-β1) induces expression of the proinflammatory and profibrotic cytokine monocyte chemoattractant protein-1 (MCP-1) in tubular epithelial cells (TECs) and thereby contributes to the tubular epithelial-mesenchymal transition (EMT), which in turn leads to the progression of tubulointerstitial inflammation into tubulointerstitial fibrosis. Exactly how TGF-β1 causes MCP-1 overexpression and subsequent EMT is not well understood. Using human tubular epithelial cultures, we found that TGF-β1 upregulated the expression of reduced nicotinamide adenine dinucleotide phosphate oxidases 2 and 4 and their regulatory subunits, inducing the production of reactive oxygen species. These reactive species activated a signaling pathway mediated by extracellular signal-regulated kinase (ERK1/2) and nuclear factor-κB (NF-κB), which upregulated expression of MCP-1. Incubating cultures with TGF-β1 was sufficient to induce hallmarks of EMT, such as downregulation of epithelial marker proteins (E-cadherin and zonula occludens-1), induction of mesenchymal marker proteins (α-smooth muscle actin, fibronectin, and vimentin), and elevated cell migration and invasion in an EMT-like manner. Overexpressing MCP-1 in cells exposed to TGF-β1 exacerbated these EMT-like changes. Pretreating cells with the antioxidant and anti-inflammatory compound arctigenin (ATG) protected them against these TGF-β1-induced EMT-like changes; the compound worked by inhibiting the ROS/ERK1/2/NF-κB pathway to decrease MCP-1 upregulation. These findings suggest ATG as a new therapeutic candidate to inhibit or even reverse tubular EMT-like changes during progression to tubulointerstitial fibrosis, and they provide the first clues to how ATG may work.
Although tumor-associated fetal protein AFP has demonstrated utility as a clinical tumor marker, the significance of intracellular AFP is still unclear. The aim of this study was to explore the role of cytoplasmic AFP during HBx induced carcinogenesis, which had not previously been recognized; 614 HCC patients were analyzed for correlation of HBV infection with AFP level, and much higher AFP levels were found in HBsAg positive patients. Tumor tissue specimens from 20 HCC patients were used for analysis of AFP and GADD45α. Analysis of HCC specimens showed that upregulation of cytoplasmic AFP is associated with down-regulation of GADD45α in neoplastic tissue. Transfected HBx promotes transcription of AFP by acting on the elements in the AFP gene regulatory region. HBx itself did not directly impact transcription of GADD45α. However, the obstruction of RAR signaling by HBx induced elevation of AFP, which led to down-regulation of GADD45α. Cytoplasmic AFP was able to interact with RAR, disrupting its entrance into the nucleus and binding to the elements in the regulatory region of the GADD45α gene. Knockdown of AFP in siRNA-transfected AFP positive cell lines was synchronously associated with an incremental increase of RAR binding to DNA, as well as upregulation of GADD45α and it was contrary in AFP gene-transfected AFP negative cell lines. These results indicate cytoplasmic AFP is not only a histochemical tumor biomarker for human hepatoma but is also an intracellular signal molecule and potential participant in HBx induced hepatocarcinogenesis.
Human urinary bladder cancer (UBC) is the fourth most common cancer and the eighth most common cause of cancer death in the USA. High mobility group box 3 (HMGB3), a member of a family of proteins containing one or more high mobility group DNA binding motifs, was reported to be overexpressed in a variety of human cancers. However, the expression and role of HMGB3 in human UBC remains unclear. Here, we found that UBC patients had upregulated HMGB at both mRNA and protein levels. Immunochemistry (IHC) evaluation of HMGB3 expression in 113 UBC clinical specimens showed that high expression of HMGB3 had positive correlation with UBC tumor size (P = 0.019), tumor WHO grade (P = 0.031), stage (P = 0.028), and lymph node metastasis (P = 0.017). Moreover, patients with higher HMGB3 expression showed a poorer overall survival rate than those with relatively low HMGB3 (P = 0.0079, log-rank test). Multivariate analysis revealed that HMGB3 expression is an independent prognostic marker. The UBC cancer cell proliferation and migration ability were measured by 3-(4, 5-dimethylthiazol-2-yl)-2, 5-diphenyltetrazolium bromide (MTT) and wound healing assays, respectively. RNA interference of HMGB3 in UBC cell lines inhibited cancer cell growth and migration, along with the downregulation of PCNA and MMP2 protein levels. In sum, our data suggests HMGB3 may serve as an important oncoprotein and indicate that overexpression of HMGB3 in UBC could be used as a potential prognostic marker.
MicroRNA-195 (miR-195) plays important roles in tumor metastasis and angiogenesis, yet its function and mechanism of action in hepatocellular carcinoma (HCC) remain to be elucidated. In this study, we aimed to confirm whether chromobox homolog 4 (CBX4) is a direct target gene of miR-195 and determine the functions of miR-195 through the CBX4 pathway. miR-195 expression was slightly lower in the HCC tissues compared with that in the adjacent normal tissues. In addition, western blotting and qRT-RCR results showed that both CBX4 mRNA and protein levels were downregulated upon miR-195 overexpression. Luciferase reporter assays revealed that CBX4 is a direct target gene of miR-195. Furthermore, overexpression of CBX4 significantly restored the proliferative, invasive and migratory capacities of the HepG2 cells. Finally, in vivo experiments confirmed that high expression of CBX4 in HepG2 cells promoted tumor growth. In conclusion, our study demonstrated that miR-195 acts as a tumor suppressor by directly targeting CBX4 in HCC. This finding suggests a potential novel strategy for therapeutic interventions of this disease.
Necroptosis is a recently discovered programmed necrosis, regulated by receptor interacting protein kinase 1 (RIP1) and RIP3 after death signal stimulation and could be specifically inhibited by necrostatin-1. The aim of this study was to investigate the role of RIP1 and RIP3 signal pathways in a mouse model of collagenase-induced intracerebral hemorrhage (ICH) and assess the effect of necrostatin-1 on brain injury after ICH. We found that RIP1 and RIP3 proteins were abundantly expressed and increased in mice brain after ICH. Necrostatin-1 pretreatment improved neurological function and attenuated brain edema in mice after ICH. Moreover, necrostatin-1 reduced RIP1-RIP3 interaction and propidium iodide (PI) positive cell death, and further inhibited microglia activation and pro-inflammatory mediator genes [tumor necrosis factor-a (TNF-α) and interleukin-1β (IL-1β)] expression after ICH. These findings indicate that RIP1/RIP3-mediated necroptosis is an important mechanism of cell death after ICH. Through inhibiting necroptosis, necrostatin-1 plays a protective role in reducing necrotic cell death after ICH. Necrostatin-1 is a promising therapeutic agent that protects cells from necroptosis and improves functional outcome.
Dysmetabolic iron overload syndrome (DIOS) is frequently observed, but the underlying mechanism remains unclear. We propose the hypothesis that hyperinsulinemia, a common characteristic of DIOS, may stimulate liver transferrin receptor 1 (TFR1) expression via the PI3K/iron regulatory protein 2 (IRP2) pathway, leading to the occurrence of DIOS. The hepatic iron content, serum iron parameters, and expressions of TFRs and IRPs in the liver were determined in rats with temporary or long-lasting hyperinsulinemia induced by acute or chronic administration of insulin. The effect of insulin on TFR1 expression and its molecular mechanism were determined in HL-7702 cells in vitro. It was found that long-lasting hyperinsulinemia significantly increased TFR1 expression in the liver and induced mild-to-moderate hepatic iron overload, which was accompanied by a normal level of serum iron. Insulin markedly upregulated both protein and mRNA levels of TFR1 in HL-7702 cells. The stability of TFR1 mRNA stability, together with expression of IRPs expression, were both significantly increased by insulin treatment. Insulin-induced TFR1 expression was blocked by IRP2, but not by IRP1 interference, and disappeared when HL-7702 cells were pretreated with LY294002, triciribine hydrate, or rapamycin. In conclusion, the findings of this study indicate that hyperinsulnemia could induce hepatic iron overload by upregulating liver TFR1 via the PI3K/AKT/mTOR/IRP2 pathway, which may be one of the main reasons for the occurrence of DIOS.
We used mice with targeted deletion of 25-hydroxyvitamin D-1 α-hydroxylase [1α(OH)ase(-/-)] to investigate whether 1,25(OH)2D3 deficiency results in male infertility mediated by 1,25(OH)2D3 or extracellular calcium and phosphorus. Male 1α(OH)ase(-/-) and their wild-type littermates fed either a normal diet or a rescue diet from weaning were mated at 6-14 wk of age with female wild-type mice on the same diet. The fertility efficiency of females was analyzed, and the reproductive phenotypes of males were evaluated by histopathological and molecular techniques. Hypocalcemic and hypophosphatemic male 1α(OH)ase(-/-) mice on a normal diet developed infertility characterized by hypergonadotropic hypogonadism, with downregulation of testicular calcium channels, lower intracellular calcium levels, decreased sperm count and motility, and histological abnormalities of the testes. The proliferation of spermatogenic cells was decreased with downregulation of cyclin E and CDK2 and upregulation of p53 and p21 expression, whereas apoptosis of spermatogenic cells was increased with upregulation of Bax and p-caspase 3 expression and downregulation of Bcl-xl expression. When serum calcium and phosphorus were normalized by the rescue diet, the defective reproductive phenotype in the male 1α(OH)ase(-/-) mice, including the hypergonadotropic hypogonadism, decreased sperm count and motility, histological abnormalities of testis, and defective spermatogenesis, was reversed. These results indicate that the infertility seen in male 1,25(OH)2D3-deficient mice is not a direct effect of active vitamin D deficiency on the reproductive system but is an indirect effect mediated by extracellular calcium and phosphorus.
Acremoniumterricola milleretal mycelium (AMM) exerts numerous protective effects on organs, and has been used in Chinese herb prescriptions to treat refractory diseases. The aim of this study was to investigate the effects of AMM on immunological hepatic fibrosis induced by porcine serum (PS) in rats. Male Sprague Dawley rats were administered 0.5 ml sterile PS by intraperitoneal injections twice a week for 18 weeks. AMM (175, 350 or 700 mg/kg) and colchicine (0.1 mg/kg) were administered intragastrically each day until the rats were sacrificed. PS administration resulted in marked hepatic fibrosis, as assessed by increased oxidative stress and hepatic collagen content, as well as α‑smooth muscle actin (α‑SMA) expression. AMM significantly reduced liver damage and fibrosis. In addition, AMM decreased the elevation in hydroxyproline, hyaluronic acid, laminin and procollagen type III; increased the activity of superoxide dismutase and glutathione peroxidase; decreased α‑SMA expression; and eliminated hepatic collagen deposits. Furthermore, AMM inhibited Smad2/3 phosphorylation and Smad7 expression. These results indicate that AMM is able to reduce oxidative stress, inhibit collagen synthesis and block the transforming growth factor‑β/Smad signaling pathway in a dose‑dependent manner.
Regulating inflammation could be an important measure for the effective treatment of cancer. Here we examine the mechanisms by which oroxylin A inhibits inflammation in RAW264.7 cells. The results demonstrate that pretreatment with oroxylin A (50, 100, and 150 μmol/L) inhibited lipopolysaccharide (LPS)-induced mRNA and protein expression of COX-2 and iNOS. In addition, oroxylin A significantly increased the protein expression of nuclear factor erythroid 2-related factor 2 (Nrf2), heme oxygenase 1 (HO-1), and NADP(H):quinone oxidoreductase (NQO1), induced Nrf2 translocation to the nucleus and up-regulated antioxidant response element (ARE)-luciferase reporter activity. Moreover, oroxylin A inhibited Nrf2 ubiquitination and proteasome activity. Transfection with Nrf2 siRNA knocked down Nrf2 expression and partially reversed oroxylin A-mediated inhibition of LPS-induced COX-2 and iNOS expression. Importantly, we showed for the first time that Nrf2 plays an important role in oroxylin A-suppressed inflammation in RAW264.7 cells. Uncovering the effect of oroxylin A on the regulation of Nrf2 signaling may be beneficial for developing new therapeutic strategies against inflammatory diseases.
Inflammation is involved in the development and/or progression of diabetic nephropathy (DN). Curcumin has been reported for its anti-inflammation activity in DN. However, the mechanisms involved in the renoprotective effects of curcumin have not been clearly demonstrated. In this study, we hypothesized that curcumin affected high glucose (HG)-induced inflammation profiles in vivo and in vitro and then prevented renal injury in diabetic rats via reversing cav-1 Tyr(14) phosphorylation that influenced TLR4 activation. Streptozotocin (STZ)-induced diabetic rats received vehicle or curcumin for twelve weeks and podocytes were treated with HG in the presence or absence of curcumin in vitro. To further evaluate the effect of cav-1 phosphorylation at Tyr(14) on HG-induced podocyte inflammation response and TLR4 activation, a recombinant plasmid GFP-Cav-1 Y14F with a mutated phosphorylation site of cav-1, was transfected into cultured podocytes. In vivo, curcumin improved histological abnormalities and fibrosis of a diabetic kidney, inhibited renal inflammatory gene expression and reduced cav-1 phosphorylation at Tyr(14) and the expression of TLR4. Pretreatment of podocytes with curcumin reduced HG-stimulated production of proinflammatory cytokines, TLR4 and the phosphorylation of cav-1. But immunohistochemistry in rat kidney showed that the elevation of TLR4 expression is more evident in the renal interstitum than in the glomerulus where podocytes are located, and the possibility that the anti-inflammatory effects of curcumin on other cells in the kidney may be mediated through the same molecular pathways as in podocytes. Our study suggests that curcumin treatment ameliorates DN via inhibition of inflammatory gene expression by reversing caveolin-1 Tyr(14) phosphorylation that influenced TLR4 activation.
Tetramethylpyrazine (TMP) is an active compound extracted from the traditional Chinese medicinal herb Chuanxiong. Recently, it has been reported that TMP enhances neurogenesis, and promotes neural stem cell differentiation toward neurons. However, its molecular basis remains unknown. Topoisomerase IIβ (TopoIIβ) is a nuclear enzyme with an essential role in neuronal development. This study aimed to investigate whether TopoIIβ is involved in TMP-induced neuronal differentiation. We examined the effect of TMP on neuronal differentiation of SH-SY5Y cells. It was found that TMP inhibited cell proliferation and induced G0/G1 cell cycle arrest. TMP promoted SH-SY5Y cells to differentiate toward post-mitotic neurons characterized by long, out-branched neurites and up-regulated neuronal markers, microtubule-associated protein 2 (MAP2) and tau. Meanwhile, we demonstrated that TopoIIβ was highly expressed following TMP treatment. To unravel how TMP affects TopoIIβ expression, two chromatin active markers, acetylated histone H3 (Ac-H3) and acetylated histone H4 (Ac-H4) were examined in this study. Our data showed that the levels of Ac-H3 and Ac-H4 were positively correlated with TopoIIβ expression in the processes of neuronal differentiation. We further performed chromatin immunoprecipitation (ChIP) analysis and identified that TMP enhanced the recruitment of Ac-H3 and Ac-H4 to the TopoIIβ gene promoter region. Therefore, we concluded that TMP may stimulate neuronal differentiation of SH-SY5Y cells through epigenetic regulation of TopoIIβ. These results suggest a novel molecular mechanism underlying TMP-promoted neuronal differentiation.
The competitive endogenous RNAs (ceRNAs) are RNA molecules that affect each other's expression through competition for their shared microRNAs (miRNAs). In this study we explored whether FOXO1 3'UTR can function as a ceRNA in repressing epithelial-to-mesenchymal transition (EMT) and metastasis of breast cancer cells via regulating miR-9 activity. We found that miR-9 binds to both the FOXO1- and E-cadherin-3'UTR, indicating that the FOXO1- and E-cadherin-3'UTR can be linked through miR-9. Follow-up analyses showed that there existed a competition of miR-9 between FOXO1 and E-cadherin-3'UTR. Thus FOXO1 3'UTR inhibits the metastases of breast cancer cells via induction of E-cadherin expression. Our results suggest that FOXO1 3'UTR may function as a miRNA-inhibitor in modulating metastasis of breast cancer cells.
The in vitro and in vivo anti-tumor efficacy of methotrexate-loaded Fe3O4-poly-L-lactide-poly(ethylene glycol)-poly-L-lactide magnetic composite microspheres (MTX-Fe3O4-PLLA-PEG-PLLA MCMs, MMCMs), which were produced by co-precipitation (C) and microencapsulation (M) in a supercritical process, was evaluated at various levels: cellular, molecular, and integrated. The results at the cellular level indicate that MMCMs (M) show a better anti-proliferation activity than raw MTX and could induce morphological changes of cells undergoing apoptosis. At the molecular level, MMCMs (M) lead to a significantly higher relative mRNA expression of bax/bcl-2 and caspase-3 than MMCMs (C) at 10 μg mL(-1) (P<0.01); and the pro-caspase-3 protein expression measured by Western blot analysis also demonstrates that MMCMs (M) can effectively activate pro-caspase-3. At the integrated level, mice bearing a sarcoma-180 tumor are used; in vivo anti-tumor activity tests reveal that MMCMs (M) with magnetic induction display a much higher tumor suppression rate and lower toxicity than raw MTX. Pharmacokinetic studies show that MMCMs (M) with magnetic induction significantly increase the accumulation of MTX in the tumor tissue compared with the other treatments. These results suggest that the MMCMs (M) prepared by the SpEDS process have great potential to play a positive role in the magnetic targeted therapy field.
IL-1β is readily detectable in numerous joint inflammations. It can change the transcriptomic signature of fibroblast-like synoviocytes (FLS) of arthritis toward promoting migration and invasion that are relevant to arthritis progression. We hypothesize that IL-1β partially contributes to the onset of osteoarthritis (OA). We compared the tissue samples from OA and fracture subjects and found that IL-1β expression was significantly higher in the OA synovium, while TNF-α expression showed no significance. We demonstrated that IL-1β significantly increases the IL-6 and IL-8 secretions of human normal FLS; however, IL-1β does not induce TNF secretion. With metabolic labeling based proteomics and pathway analysis, we found that IL-1β significantly increases the TNF downstream protein expression in FLS even with complete absence of TNF and/or blocking of the NF-κB pathway. Among these proteins, we verified that p62 can differentiate the OA from fracture synovitis. In conclusion, we demonstrated that IL-1β can amplify the TNF downstream protein signals in human synoviocytes in a TNF-independent manner; in addition, p62 is a potential FLS biomarker for synovitis.
The NEDD8-activating enzyme (NAE) is an emerging target for cancer therapy, which regulates the degradation and turnover of a variety of cancer-related proteins by activating the cullin-RING E3 ubiquitin ligases. Among a limited number of known NAE inhibitors, the covalent inhibitors have demonstrated the most potent efficacy through their covalently linked adducts with NEDD8. Inspired by this unique mechanism, in this study, a novel combined strategy of virtual screening (VS) was adopted with the aim to identify diverse covalent inhibitors of NAE. To be specific, a docking-enabled pharmacophore model was first built from the possible active conformations of chosen covalent inhibitors. Meanwhile, a dynamic structure-based phamacophore was also established based on the snapshots derived from molecular dynamic simulation. Subsequent screening of a focused ZINC database using these pharmacophore models combined with covalent docking discovered three novel active compounds. Among them, compound LZ3 exhibited the most potent NAE inhibitory activity with an IC50 value of 1.06 ± 0.18 μM. Furthermore, a cell-based washout experiment proved the proposed covalent binding mechanism for compound LZ3, which confirmed the successful application of our combined VS strategy, indicating it may provide a viable solution to systematically discover novel covalent ligands.
Cisplatin is one of the major chemotherapeutic agents used against different human cancers. A better understanding of the downstream cellular targets of cisplatin will provide information on its mechanism of action. FOXO3a is a member of the FOXO transcription factor family, which modulates the expression of genes involved in cell cycle arrest, apoptosis, and other cellular processes. In this study, we have investigated the effects of cisplatin in a panel of lung cancer cell lines. The results showed that cisplatin inhibited the proliferation of these lung cancer cell lines by inhibiting the PI3K/AKT pathway, with evidence of decreasing phosphorylation of PI3K and AKT under cisplatin treatment, and constitutively activating AKT1 could reduce cisplatin-induced cell apoptosis. More importantly, cisplatin significantly inhibited FOXO3a phosphorylation (at Thr32, AKT phosphorylation site) and induced FOXO3a nuclear accumulation, which in turn increased the expression of FOXO3a-dependent apoptotic protein Bim. Knockdown of FOXO3a expression using small interfering RNA attenuated cisplatin-induced apoptosis. Furthermore, activation of FOXO3a induced cell apoptosis irrespective of p53 status, whereas p53 may act as FOXO3a downstream molecules involved in cisplatin-induced cell apoptosis. Together, our findings suggested that FOXO3a is a relevant mediator of the cytotoxic effects of cisplatin in lung cancer cells.
Essential oils of Cymbopogon citratus were already reported to have wide ranging medical and industrial applications. However, information on polysaccharides from the plant and their anticancer activities are limited. In the present study, polysaccharides from C. citratus were extracted and fractionated by anion exchange and gel filtration chromatography. Two different polysaccharide fractions such as F1 and F2 were obtained, and these fractions were found to have distinct acidic polysaccharides as characterized by their molecular weight and sugar content. NMR spectral analysis revealed the presence of (1→4) linked b-d-Xylofuranose moiety in these polysaccharides. Using these polysaccharide fractions F1 and F2, anti-inflammatory and anticancer activities were evaluated against cancer cells in vitro and the mechanism of action of the polysaccharides in inducing apoptosis in cancer cells via intrinsic pathway was also proposed. Two different reproductive cancer cells such as Siha and LNCap were employed for in vitro studies on cytotoxicity, induction of apoptosis and apoptotic DNA fragmentation, changes in mitochondrial membrane potential, and profiles of gene and protein expression in response to treatment of cells by the polysaccharide fractions. These polysaccharide fractions exhibited potential cytotoxic and apoptotic effects on carcinoma cells, and they induced apoptosis in these cells through the events of up-regulation of caspase 3, down-regulation of bcl-2 family genes followed by cytochrome c release.
Marine macroalgae consist of a range of bioactive molecules exhibiting different biological activities, and many of these properties are attributed to sulfated polysaccharides, fucoxanthin, phycobiliproteins, and halogenated compounds. In this study, a glycoprotein (GLP) with a molecular mass of ∼48 kDa was extracted and purified from Codium decorticatum and investigated for its cytotoxic properties against human MDA-MB-231 breast cancer cells. The IC₅₀ values of GLP against MDA-MB-231 and normal breast HBL-100 cells (control) were 75 ± 0.23 μg/mL (IC₂₅), 55 ± 0.32 μg/mL (IC₅₀), and 30 ± 0.43 μg/mL (IC₇₅) and 90 ± 0.57 μg/mL (IC₂₅), 80 ± 0.48 μg/mL (IC₅₀), and 60 ± 0.26 μg/mL (IC₇₅), respectively. Chromatin condensation and poly(ADP-ribose) polymerase (PARP) cleavage studies showed that the GLP inhibited cell viability by inducing apoptosis in MDA-MB-231 cells. Induction of mitochondria-mediated intrinsic apoptotic pathway by GLP was evidenced by the events of loss of mitochondrial membrane potential (ΔΨ(m)), bax/bcl-2 dysregulation, cytochrome c release, and activation of caspases 3 and 9. Apoptosis-associated factors such as reactive oxygen species (ROS) formation and loss of ΔΨ(m) were evaluated by DCFH-DA staining and flow cytometry, respectively. Cell cycle arrest of G₂/M phase and expression of apoptosis associated proteins were determined using flow cytometry and Western blotting, respectively.
MicroRNAs have recently emerged as key regulators of gastric cancers. Here we found that miR-145, miR-133a and miR-133b were down-regulated in gastric cancer tissues and cell lines. Overexpression of miR-145, miR-133a and miR-133b induced G1 cell cycle arrest and inhibited cell proliferation, migration and invasion in vitro. MiR-145, miR-133a and miR-133b targeted the transcription factor SP1, knockdown of which reduced the expression of MMP-9 and Cyclin D1 that were involved in cell growth and invasion. Thus, our findings demonstrated for the first time that miR-145, miR-133a and miR-133b suppressed the proliferation, migration, invasion and cell cycle progression of gastric cancer cells through decreasing expression of Sp1 and its downstream proteins.
Inflammation is an important contributor to the development of Alzheimer's disease (AD). Anti-inflammatory medication may offer promising treatment for AD. Hydroxy-safflor yellow A (HSYA), a chemical component of the safflower yellow pigments, has been reported to exert potent immunosuppressive effects. This study examined the anti-inflammatory effects of HSYA in Aβ₁₋₄₂-treated BV-2 microglia cells. The mRNA levels of IL-1β, IL-4, IL-10, TNF-α, COX-2 and iNOS were detected by real-time PCR. Western blotting was used to determine the protein expression of COX-2, TNF-α, iNOS, Janus Kinase 2 (JAK2), p-JAK2, signal transducers and activators of transcription 3 (STAT3) and p-STAT3. BV2-conditioned medium was used to treat SH-SY5Y cells and primary neuronal cells in indirect toxicity experiments. Cell viability and apoptosis were assessed using MTT assay and Annexin V/PI staining respectively. The results demonstrated that HSYA significantly reduced the expression of the pro-inflammatory mediators and inhibited Aβ₁₋₄₂-induced neuroinflammation. Moreover, HSYA protected primary cortical neurons and SH-SY5Y cells against microglia-mediated neurotoxicity. HSYA also enhanced the phosphorylation of JAK2/STAT3 pathway and inhibition of JAK2 by AG 490 attenuated the anti-inflammatory effects of HSYA. Overall, our findings suggested that HSYA inhibited Aβ₁₋₄₂-induced inflammation and conferred neuroprotection partially through JAK2/STAT3 pathway, indicating that HSYA could be a potential drug for the treatment of AD.
Ubiquitin C-terminal hydrolase L1 (UCH-L1) is abundantly expressed in the brain and is critical for the normal function of synapses. cAMP response element binding protein (CREB) is a transcription factor which initiates the expression of proteins that related to the regulation of synaptic plasticity and memory function. Studies have shown that UCH-L1 can influence the expression and activity of CREB, but the underlying mechanisms remain unclear. In this study, we used UCH-L1 inhibitor LDN to treat mice hippocampal slices and found that UCH-L1 inhibition caused the dephosphorylation of CREB at Ser133 site. Meanwhile, hyperphosphorylation of microtubule-associated protein tau; increased expression of synaptic protein components of PSD-95 and synapsin-1, and decreased activity of tyrosine kinase Fyn were observed after UCH-L1 inhibition. Moreover, all these alternations have an influence on the normal function of N-methyl-D-aspartate (NMDA) receptor NR2B subunit which is likely to result in the dephosphorylation of CREB. We also found that LDN treatment mediated protein kinase A (PKA) deactivation was involved in the dephosphorylation of CREB. Thus, our study introduces a novel possible mechanism for elaborating the effects of UCH-L1 inhibition on the CREB activity and the implicated signaling pathways.
Angiotensin II (Ang II)-elicited excessive proliferation, hypertrophy and migration of vascular smooth muscle cells (VSMCs) are vital to the pathogenesis of atheroclerosis. Glutathione S-transferase pi (GSTpi) exists extensively in various kinds of cells and protects cells against different stresses. However, knowledge remains limited about what GSTpi acts in VSMCs. We investigated the effect of GSTpi on Ang II-induced VSMC proliferation, hypertrophy and migration and its latent mechanism. Overexpression and RNAi experiments demonstrated that GSTpi inhibited Ang II-induced proliferation, hypertrophy and migration of VSMCs and arrested progression of cell cycle from G0/G1 to S phase. Immunoprecipitation, mass spectrometry and confocal microscopy analyses showed that GSTpi directly associated with signal transducer and activator of transcription 3 (STAT3) to prevent Ang II-triggered binding of Src to STAT3 and thus suppressed Ang II-stimulated phosphorylation and nuclear translocation of STAT3, as well as cyclin D1 expression. In contrast, GSTpi didn't affect Ang II-activated extracellular signal-regulated kinase (ERK1/2). GSTpi acts as a negative regulator to prevent Ang II-triggered proliferative signaling in VSMCs, suggesting that it may protect vessels against the stresses associated with atherosclerosis formation.
Hepatic stellate cells (HSCs) are liver-specific pericytes that are recruited to vessels and secret pro-angiogenic cytokines, and thus actively involved in pathological vascularization during liver fibrosis. Peroxisome proliferator-activated receptor-γ (PPARγ) is a switch molecule controlling HSC activation. We investigated PPARγ regulation of angiogenic signal transduction and the molecular mechanisms involved in HSCs. Primary rat HSCs and liver sinusoidal endothelial cells (LSECs) were isolated and used in this study. Boyden chamber and tubulogenesis assays, identified that focal adhesion kinase (FAK)-RhoA signaling activated by platelet-derived growth factor (PDGF) was required for HSC motility and the associated vascularization. PDGF also stimulated vascular endothelial growth factor (VEGF) expression and HSC-driven vascularization through signals mediated by extracellular signal-regulated kinase (ERK) and mammalian target of rapamycin (mTOR). Gain- and loss-of-function analyses demonstrated that activation of PPARγ interrupted FAK-RhoA, ERK and mTOR cascades and inhibited HSC-based vascularization. Molecular evidence further revealed that PPARγ attenuation of HSC angiogenic properties was dependent on inhibition of PDGF-β receptor expression. We concluded that PPARγ inhibited angiogenic signal transduction through transrepression of PDGF-β receptor leading to reduced HSC motility, reduced VEGF expression, and thereby attenuated HSC-driven angiogenesis. PPARγ could be a molecular target for preventing vascular remolding in hepatic fibrosis.
There are contradictory results about the effect of angiotensin-converting enzyme inhibitors (ACEIs) on bone. This study was performed to address the skeletal renin-angiotensin system (RAS) activity and the effects of the ACEI, captopril, on the bone of streptozotocin-induced type 1 diabetic mice. Histochemical assessment on bone paraffin sections was conducted by Safranin O staining and tartrate-resistant acid phosphatase staining. Micro-computed tomography was performed to analyze bone biological parameters. Gene and protein expression were determined by real-time polymerase chain reaction and immunoblotting, respectively. Type 1 diabetic mice displayed osteopenia phenotype and captopril treatment showed no osteoprotective effects in diabetic mice as shown by the reduction of bone mineral density, trabecular thickness and bone volume/total volume. The mRNA expression of ACE and renin receptor, and the protein expression of renin and angiotensin II were markedly up-regulated in the bone of vehicle-treated diabetic mice compared to those of non-diabetic mice, and these molecular changes of skeletal RAS components were effectively inhibited by treatment with captopril. However, treatment with captopril significantly elevated serum tartrate-resistant acid phosphatase 5b levels, reduced the ratio of osteoprotegerin/receptor activator of nuclear factor-κB ligand expression, increased carbonic anhydrase II mRNA expression and the number of matured osteoclasts and decreased transforming growth factor-β and osteocalcin mRNA expression in the tibia compared to those of diabetic mice. The present study demonstrated that the use of the ACEI, captopril, has no beneficial effect on the skeletal biological properties of diabetic mice. However, this could be attributed, at least partially, to its suppression of osteogenesis and stimulation of osteoclastogenesis, even though it could effectively inhibit high activity of local RAS in the bone of diabetic mice.
This study was undertaken to identify growth hormone (GH) responsive proteins and protein expression patterns by short-term recombinant human growth hormone (rhGH) therapy in patients with idiopathic short stature (ISS) using proteomic analysis. Seventeen children (14 males and three females) with ISS were included. They were treated with rhGH at a dose of 0.31 ± 0.078 mg/kg/week for 3 months. Immunodepletion of six highly-abundant serum proteins followed by 2D DIGE analysis, and subsequent MALDI TOF MS, were employed to generate a panel of proteins differentially expressed after short-term rhGH therapy and verify the differences in serum levels of specific proteins by rhGH therapy. Fourteen spots were differentially expressed after rhGH treatment. Among them, apo E and apo L-1 expression were consistently enhanced, whereas serum amyloid A was reduced after rhGH therapy. The differential expressions of these proteins were subsequently verified by Western blot analysis using sera of the before and after rhGH treatment. This study suggests that rhGH therapy influences lipoprotein metabolism and enhances apo L-1 protein expression in ISS patients.
A high dose of granulocyte colony stimulating factor (G-CSF) is widely used to mobilize hematopoietic stem and progenitor cells (HSPC), but G-CSF is relatively inefficient and may cause adverse effects. Recently, adrenaline has been found to play important roles in HSPC mobilization. In this study, we explored whether adrenaline combined with G-CSF could induce HSPC mobilization in a mouse model. Mice were treated with adrenaline and either a high or low dose of G-CSF alone or in combination. Peripheral blood HSPC counts were evaluated by flow cytometry. Levels of bone marrow SDF-1 were measured by ELISA, the transcription of CXCR4 and SDF-1 was measured by real-time RT-PCR, and CXCR4 protein was detected by Western blot. Our results showed that adrenaline alone fails to mobilize HSPCs into the peripheral blood; however, when G-CSF and adrenaline are combined, the WBC counts and percentages of HSPCs are significantly higher compared to those in mice that received G-CSF alone. The combined use of adrenaline and G-CSF not only accelerated HSPC mobilization, but also enabled the efficient mobilization of HSPCs into the peripheral blood at lower doses of G-CSF. Adrenaline/G-CSF treatment also extensively downregulated levels of SDF-1 and CXCR4 in mouse bone marrow. These results demonstrated that adrenaline combined with G-CSF can induce HSPC mobilization by down-regulating the CXCR4/SDF-1 axis, indicating that the use of adrenaline may enable the use of reduced dosages or durations of G-CSF treatment, minimizing G-CSF-associated complications.
Bmal1 is a transcription factor that plays a central role in the regulation of circadian rhythms. Recent study reported that Bmal1-/- mice displayed many known features of premature ageing, such as reduction of bone mass. Our previous study has found that both the proliferation of bone marrow mesenchymal stem cells (BMSCs) and Bmal1 expression decreased with advancing age. It seemed that a positive correlation existed between Bmal1 protein level and the proliferative activity of BMSCs. β-catenin, the core factor of the canonical Wnt pathway, also showed reduced expression in aged mice. In order to further confirm this, we constructed a lentiviral vector to over-express Bmal1 in NIH-3T3 cells; successful transfection was verified. The cell proliferation rate of infected cells was higher than the non-transfected NIH-3T3 cells, suggesting that circadian clock gene Bmal1 can promote proliferation. β-catenin showed an increased expression in NIH-3T3 cells after Bmal1 over-expression, indicating that activation of the canonical Wnt pathway might be the mechanism underlying the effect of circadian clock gene Bmal on promoting cell proliferation.
Cholestasis has limited therapeutic options and is associated with high morbidity and mortality. The A(1) adenosine receptor (A(1)AR) was postulated to participate in the pathogenesis of hepatic fibrosis induced by experimental extrahepatic cholestasis; however, the contribution of A(1)AR to intrahepatic cholestatic liver injury remains unknown. Here, we found that mice lacking A(1)AR were resistant to alpha-naphthyl isothiocyanate (ANIT)-induced liver injury, as evidenced by lower serum liver enzyme levels and reduced extent of histological necrosis. Bile acid accumulation in liver and serum was markedly diminished in A(1)AR(-/-) mice compared with wild-type (WT) mice. However, biliary and urinary outputs of bile acids were significantly enhanced in A(1)AR(-/-) mice. In the liver, mRNA expression of genes related to bile acid transport (Bsep and Mdr2) and hydroxylation (Cyp3a11) was increased in A(1)AR(-/-) mice. In the kidney, A(1)AR deficiency prevented the decrease of glomerular filtration rate caused by ANIT. Treatment of WT mice with A(1)AR antagonist DPCPX also protected against ANIT hepatotoxicity. Our results indicated that lack of A(1)AR gene protects mice from ANIT-induced cholestasis by enhancing toxic biliary constituents efflux through biliary excretory route and renal elimination system and suggested a potential role of A(1)AR as therapeutic target for the treatment of intrahepatic cholestasis.
Surgery often leads to neuroinflammation, which mainly acts as the activation of microglia cells. Propofol is always used for induction and maintenance of anesthesia prior to surgical trauma, whereas whether or not it could attenuate neuroinflammation used prophylactically is not well defined. In the present study, we incubated BV-2 microglia cells with 1 μg/ml lipopolysaccharide (LPS) to mimic neuroinflammation in vitro. Firstly, cell viability was measured using 3-(4,5-dimethylthiazol-2-yl)-2, 5-diphenyltetrazolium bromide (MTT) assay and the data indicated that propofol would not reduce cell viability unless its concentration reached 300 μM. Secondly, BV-2 microglia cells were pretreated with 30 μM propofol (clinically relevant concentration), and then stimulated with LPS. The results showed that the production of tumor necrosis factor-α (TNF-α), interleukin (IL)-1β and IL-10 was considerably increased by LPS, but the change could be markedly attenuated by pretreatment with propofol. Meanwhile, pretreatment with propofol inhibited LPS-induced augmentation of toll-like receptor 4 (TLR4) expression at both mRNA and protein levels and further upregulated LPS-induced inactivation of glycogen synthase kinase-3β (GSK-3β) in BV-2 microglia cells. These results indicated, at least in part, that pretreatment with propofol can protect BV-2 microglia cells against LPS-induced inflammation. Downregulation of TLR4 expression and inactivation of GSK-3β may be involved in its protective effect.
Autophagy is a tightly-regulated catabolic process that involves the degradation of intracellular components via lysosomes. Although the pivotal role of autophagy in cell growth, development, and homeostasis has been well understood, its function in cancer prevention and intervention remains to be delineated. The aim of this study was to investigate the function and mechanism of autophagy induced by oroxylin A, a natural mono-flavonoid extracted from Scutellariae radix. We found for the first time that oroxylin A induced Beclin 1-mediated autophagy in human hepatocellular carcinoma HepG2 cells. Time-lapse video microscopy and western blotting studies showed that treatment of cells with 80 μM oroxylin A resulted in the conversion of water soluble MAP-LC3 (LC3-I) to the lipidated and autophagosome-associated form (LC3-II) after 12hours; then autophagosome-lysosome fusion and lysosome degradation after 24 hours was required in oroxylin A-mediated cell death. This induction was associated with the suppressing of PI3K-PTEN-Akt-mTOR signaling pathway by oroxylin A. Our results also showed that autophagy took place before noticeable apoptosis can be observed. It was further demonstrated that oroxylin A-triggered autophagy contributed to cell death using over-expression of autophagy-related gene (Atg5 and Atg7) and inhibition of autophagy by siBeclin 1 and 3-methyladenine (3-MA). In vivo study, oroxylin A inhibited xenograft tumor growth and induced obvious autophagy in tumors. Taken together, we conclude that oroxylin A exhibits autophagy-mediated antitumor activity in a dose and time-dependent manner in vivo and in vitro. These findings define and support a novel function of autophagy in promoting death of hepatocellular carcinoma cells.
P-glycoprotein (P-gp) is an ATP-dependent efflux transporter highly expressed in gastrointestinal tract and multidrug resistance tumor cells. Inhibition or induction of P-gp can cause drug-drug interactions and thus influence the effects of P-gp substrate drugs. Previous studies indicated that 20(S)-ginsenoside Rh2 [20(S)-Rh2] could synergistically enhance the anticancer effects of conventional chemotherapeutic agents at a nontoxic dose. The aim of present study was to investigate in vitro and in vivo whether 20(S)-Rh2 was a P-gp inhibitor and analyze the possible inhibitory mechanisms and potential herb-drug interactions. Results showed that in vitro, 20(S)-Rh2 significantly enhanced rhodamine 123 retention in cells and decreased the efflux ratio of digoxin, fexofenadine, and etoposide, which were comparable to the effects of the established P-gp inhibitor verapamil. However, the transport of 20(S)-Rh2 suggested that 20(S)-Rh2 was not a P-gp substrate. Furthermore, the inhibitory effect persisted for at least 3 h after removal of 20(S)-Rh2. Unlike P-gp substrates, 20(S)-Rh2 inhibited both basal and verapamil-stimulated P-gp ATPase activities. It also significantly decreased UIC2 binding fluorescence, a marker for conformational change of P-gp. In situ and in vivo experiments showed that 20(S)-Rh2 increased the area under the plasma concentration-time curve and maximum plasma concentration of digoxin, fexofenadine, and etoposide significantly without affecting terminal elimination half-time. Long-term treatment with 20(S)-Rh2 failed to affect intestinal P-gp expression in vitro and in vivo. In conclusion, 20(S)-Rh2 is a potent noncompetitive P-gp inhibitor, which indicates a potential herb-drug interaction when 20(S)-Rh2 is coadministered with P-gp substrate drugs. It could increase the absorption of P-gp substrate drugs without long-term induction of P-gp expression in rats.
MicroRNAs (miRNAs) are short noncoding RNA molecules, which posttranscriptionally regulate genes expression and play crucial roles in diverse biological processes, such as development, differentiation, apoptosis and proliferation. Here, we investigated the possible role of miRNAs in the development of multidrug resistance (MDR) in human gastric and lung cancer cell lines. We found that miR-181b was downregulated in both multidrug-resistant human gastric cancer cell line SGC7901/vincristine (VCR) and multidrug-resistant human lung cancer cell line A549/cisplatin (CDDP), and the downregulation of miR-181b in SGC7901/VCR and A549/CDDP cells was concurrent with the upregulation of BCL2 protein, compared with the parental SGC7901 and A549 cell lines, respectively. In vitro drug sensitivity assay demonstrated that overexpression of miR-181b sensitized SGC7901/VCR and A549/CDDP cells to anticancer drugs, respectively. The luciferase activity of a BCL2 3'-untranslated region-based reporter construct in SGC7901/VCR and A549/CDDP cells suggests that a new target site in the 3'UTR of BCL2 of the mature miR-181s (miR-181a, miR-181b, miR-181c and miR-181d) was found. Enforced miR-181b expression reduced BCL2 protein level and sensitized SGC7901/VCR and A549/CDDP cells to VCR-induced and CDDP-induced apoptosis, respectively. Taken together, our findings suggest that miR-181b could play a role in the development of MDR in both gastric and lung cancer cell lines, at least in part, by modulation of apoptosis via targeting BCL2.
BACKGROUND:
Isoflurane disrupts brain development of neonatal mice, but its mechanism is unclear. We explored whether isoflurane damaged developing hippocampi through FASL-FAS signaling pathway, which is a well-known pathway of apoptosis.
METHOD:
Wild type and FAS- or FASL-gene-knockout mice aged 7 days were exposed to either isoflurane or pure oxygen. We used western blotting to study expressions of caspase-3, FAS (CD95), and FAS ligand (FASL or CD95L) proteins, TUNEL staining to count apoptotic cells in hippocampus, and Morris water maze (MWM) to evaluate learning and memory.
RESULT:
Isoflurane increased expression of FAS and FASL proteins in wild type mice. Compared to isoflurane-treated FAS- and FASL-knockout mice, isoflurane-treated wild type mice had higher expression of caspase-3 and more TUNEL-positive hippocampal cells. Expression of caspase-3 in wild isoflurane group, wild control group, FAS/FASL-gene-knockout control group, and FAS/FASL-gene-knockout isoflurane group showed FAS or FASL gene knockout might attenuate increase of caspase-3 caused by isoflurane. MWM showed isoflurane treatment of wild type mice significantly prolonged escape latency and reduced platform crossing times compared with gene-knockout isoflurane-treated groups.
CONCLUSION:
Isoflurane induces apoptosis in developing hippocampi of wild type mice but not in FAS- and FASL-knockout mice and damages brain development through FASL-FAS signaling.
RATIONALE:
Numerous studies have demonstrated that neuroinflammation is associated with depression-like symptoms and neuropsychological disturbances, and cysteinyl leukotriene receptor 1 (CysLT1R) was reported to be involved in neuroinflammation. The pathophysiological role of CysLT1R has been reported in several types of brain damage. However, the role of CysLT1R in depression remains to be elucidated.
OBJECTIVES:
We aimed to investigate the effect of hippocampal CysLT1R downregulation on depressive behaviors and neuroinflammatory responses in mice exposed to chronic mild stress (CMS).
RESULTS:
We firstly found that expression of hippocampal CysLT1R was gradually increased over CMS exposure, while 3 weeks treatment with fluoxetine reversed the increment of hippocampal CysLT1R expression. Hippocampal CysLT1R knockdown suppressed CMS-induced depressive-like behaviors as evidenced by decreases in immobility time in tail suspension test (TST), decreased latency to feed in novelty-suppressed feeding (NSF) test, and by increase in the number of entries and decrease in time spent in the open arm in elevated plus maze (EPM) test. Increments of hippocampal NF-κB p65, IL-1β, and TNF-α induced by CMS were also prevented by hippocampal CysLT1R knockdown beforehand.
CONCLUSIONS:
Hippocampal CysLT1R participates in depression, and knockdown of hippocampal CysLT1R prevents CMS-induced depressive-like behaviors and neuroinflammation, suggesting that suppression of CysLT1R could prevent the development of depression.
OBJECTIVE:
To investigate the signal pathway of honokiol-induced apoptosis in H4 human neuroglioma cells and to evaluate whether p53 signaling and cell cycle arrest were involved in honokiol-treated H4 human neuroglioma cells.
METHODS:
The cell viability was detected by the CCK8 assay. The cell apoptosis was assessed by annexin V-PI double-labeling staining and hoechst 33342 staining. The protein expression of cell cycle regulators and tumor suppressors were analyzed by western blotting.
RESULTS:
Treatment of H4 human neuroglioma cells with honokiol induced cell death in a dose-and time-dependent manner by using CCK8 assay. Consistent with the CCK8 assay, the flow cytometry results showed that the proportion of the apoptosis cells increased after honokiol when compared with untreated group. Moreover, H4 human neuroglioma cells exposed to honokiol, resulted in an accumulation of cells in S and G2/M phase. Apoptotic bodies were clearly observed in human neuroglioma cells when treated with honokiol and then stained with Hoechst 33342. The expression of Cyclin B1, CDC2 and cdc25C were downregulated, however, the expression of p-CDC2 and p-cdc25c was significantly upregulated when the neuroglioma cells were exposed to honokiol. Moreover, p53, p21 and Bax/Bcl-2 were significantly upregulated by honokiol treatment.
CONCLUSIONS:
These results confirmed that honokiol could induce apoptosis in human neuroglioma cells, the underlying molecular mechanisms, at least partially, through activation p53 signaling and induction of cell cycle arrest.
BACKGROUND:
The microRNA miR-101 is downregulated in several cancers, including bladder cancer. However, miR-101's role in the invasion, metastasis, and chemosensitivity of bladder cancer cells remains unclear. This study was conducted to determine miR-101's role on the lymphangiogenic molecule vascular endothelial growth factor C (VEGF-C) and their effects upon bladder cancer cell migration, invasion, and chemosensitivity to cisplatin.
METHODS:
Two bladder cancer cell lines (T24 and 5637) and the tool cell line 293T were employed here. Bladder cancer cells were transfected with either a miR-101 overexpression vector or a scrambled-sequence lentivirus, both of which exhibited a high transfection efficiency. Non-transfection was used as a mock negative control. Wound healing and Transwell assays were performed to measure cell migration and invasiveness. A luciferase reporter assay was performed to validate miR-101's interaction with VEGF-C's 3' untranslated region followed by RT-PCR and Western blot confirmation. An MTS assay was used to evaluate the cisplatin sensitivity of the cell lines.
RESULTS:
miR-101 overexpression significantly inhibited the migration and invasiveness while significantly enhancing cisplatin sensitivity. miR-101 negatively regulated VEGF-C protein expression, and VEGF-C overexpression rescued the effects of miR-101 overexpression, indicating that miR-101 negatively regulates VEGF-C protein expression post-transcriptionally. miR-101 and VEGF-C interference independently enhanced cisplatin cytotoxicity in bladder cancer cells.
CONCLUSIONS:
miR-101 suppresses VEGF-C expression, inhibits cell migration and invasion, and increases cisplatin sensitivity in bladder cancer cells. This study provides new insight into miR-101's role in bladder cancer and shows miR-101's promise as a potential molecular target for bladder cancer.
OBJECTIVE:
This study was performed to investigate PTX3-mediated iNOS expression and IKK/IκB/NF-κB activation in PA-induced atherosclerotic HUVECs injury model.
METHODS:
The cell viability was detected by the CCK8 assay. The cell apoptosis was assessed by annexin V-PI double-labeling staining. Expression of genes and proteins were analyzed by real-time PCR and western blotting respectively. Cells were transfected with siRNAs as a gene silencing methods.
RESULTS:
PA induced cell apoptosis in human umbilical vein endothelial cells in a time and dose-dependent manner. PA also induced upregulation expression of PTX3. TPCA-1, an inhibitor of IKK-2, could suppress the expression of PTX3 and phospho-IκB-α in PA-induced endothelial dysfunction cell model. We also found that transfection of cells with PTX3 siRNA reduced the expression of iNOS and NO, and protected PA-induced cell apoptosis in HUVECs.
CONCLUSIONS:
PTX3 could exacerbate endothelial dysfunction, at least partially, through IKK/IκB/NF-κB activation and overexpression of iNOS and NO, and advance the development of atherosclerosis.
OBJECTIVE:
Long non-coding RNAs (lncRNAs) play important roles in diverse biological processes, such as transcriptional regulation, cell growth and tumorigenesis. However, little was known about whether lncRNA HIF 1 alpha-antisense RNA 1 (HIF1a-AS1) in regulating the proliferation and apoptosis of VSMCs in vitro and the expression of HIF1a-AS1 in serum of TAA patients.
METHODS:
The cell viability was detected by the CCK8 assay. The cell apoptosis was assessed by annexin V-PI double-labeling staining. Expression of genes and proteins were analyzed by real-time PCR and western blotting respectively. Cells were transfected with siRNAs as a gene silencing methods.
RESULTS:
In serum of TAA patients, the expression of HIF1a-AS1 was significantly increased (superior to 6 folds) compared to the normal control. Moreover, PA induced cell apoptosis in VSMCs in a time- and dose-dependent manner, and the proportion of the apoptotic cells had gained as compared to untreatment group. PA also induced upregulation expression of HIF1a-AS1. We also found that transfection of cells with HIF1a-AS1 siRNA decreased the expression of caspase3 and caspase8 and increased the expression of Bcl2, and protected PA-induced cell apoptosis in VSMCs.
CONCLUSIONS:
HIF1a-AS1 was overexpressed in the thoracoabdominal aorta aneurysm and the interaction between HIF1a-AS1 and apoptotic proteins plays a key role in the proliferation and apoptosis of VSMCs in vitro, which may contribute to the pathogenesis of thoracoabdominal aorta aneurysm.
Luteolin has recently been proven to exert neuroprotection in a variety of neurological diseases; however, its roles and the underlying mechanisms in traumatic brain injury are not fully understood. The present study was aimed to investigate the neuroprotective effects of luteolin in models of traumatic brain injury (TBI) and the possible role of the Nrf2-ARE pathway in the putative neuroprotection. A modified Marmarou׳s weight-drop model in mice and the scratch model in mice primary cultured neurons were used to induce TBI. We determined that luteolin significantly ameliorated secondary brain injury induced by TBI, including neurological deficits, brain water content, and neuronal apoptosis. Furthermore, the level of malondialdehyde (MDA) and the activity of glutathione peroxidase (GPx) were restored in the group with luteolin treatment. in vitro studies showed that luteolin administration lowered the intracellular reactive oxygen species (ROS) level and increased the neuron survival. Moreover, luteolin enhanced the translocation of Nrf2 to the nucleus both in vivo and in vitro, which was proved by the results of Western blot, immunohistochemistry, and electrophoretic mobility shift assay (EMSA). Subsequently upregulation of the expression of the downstream factors such as heme oxygenase 1 (HO1) and
NAD(P)H:
quinone oxidoreductase 1 (NQO1) was also examined. However, luteolin treatment failed to provide neuroprotection after TBI in Nrf2(-/-) mice. Taken together, these in vivo and in vitro data demonstrated that luteolin provided neuroprotective effects in the models of TBI, possibly through the activation of the Nrf2-ARE pathway.
Copyright © 2014 Elsevier Inc. All rights reserved.
OBJECTIVES:
miR-181a is involved in immunity, metabolism, tumor suppression or carcinogenesis reported by many other studies. However, its role in the development of chemosensitivity to adriamycin in low-invasive breast cancer cells remains unclear. The aim of this study is to define the function role of miR-181a in promoting the efficacy of adriamycin-based neoadjuvant chemotherapy.
METHODS:
Cell survival analysis was detected by Cell Counting Kit-8 assay. Apoptotic cells were quantitatively detected using FITC Annexin V apoptosis Detection Kit I. Bcl-2 protein expression was measured by western blot. Luciferase reporter vector with the putative BCL-2 3' untranslated region (3'UTR) was constructed to explore whether BCL-2 was a direct target gene of miR-181a. Real-time PCR was performed to test the expression of miR-181a and Bcl-2 in the selected breast cancer tissue samples.
RESULTS:
The down-regulation of miR-181a decreased adriamycin-induced apoptosis in MCF-7 cells. Transfected with miR-181a mimic in cells resulted in the decreased expression of Bcl-2. The alteration of miR-181a expression did not significantly affect the chemosensitivity to adriamycin in MCF-7 and MCF-7/ADR cells with genetic knockout of Bcl-2. miR-181a may suppress Bcl-2 expression by forming imperfect base pairing with the 3'UTR of Bcl-2 gene such that a negative relationship between miR-181a and Bcl-2 in MCF-7 and MCF-7/ADR cells is observed.
CONCLUSIONS:
At least in part, the detection of miR-181a may direct the clinical medication in patients with neoadjuvant chemotherapy because of miR-181a enhanced adriamycin-induced apoptosis via targeting Bcl-2.
© 2013 S. Karger AG, Basel.
OBJECTIVE:
This study was designed to investigate the effects of mineral trioxide aggregate (MTA) on the osteo/odontogenic differentiation of inflammatory dental pulp stem cells (iDPSCs).
MATERIALS AND METHODS:
inflammatory DPSCs were isolated from the inflammatory pulps of rat incisors and cocultured with MTA-conditioned medium. MTT assay and flow cytometry were performed to evaluate the proliferation of iDPSCs. Alkaline phosphatase (ALP) activity, alizarin red staining, real-time RT-PCR, and Western blot assay were used to investigate the differentiation capacity as well as the involvement of NF-κB pathway in iDPSCs.
RESULTS:
Mineral trioxide aggregate-treated iDPSCs demonstrated the higher ALP activity and formed more mineralized nodules than the untreated group. The odonto/osteoblastic markers (Alp, Runx2/RUNX2, Osx/OSX, Ocn/OCN, and Dspp/DSP, respectively) in MTA-treated iDPSCs were significantly upregulated as compared with untreated iDPSCs. Mechanistically, cytoplastic phos-P65 and nuclear P65 in MTA-treated iDPSCs were significantly increased in a time-dependent manner. Moreover, the inhibition of NF-κB pathway suppressed the MTA-induced odonto/osteoblastic differentiation of iDPSCs, as indicated by decreased ALP levels, weakened mineralization capacity and downregulated levels of odonto/osteoblastic genes (Osx, Ocn, and Dspp).
CONCLUSIONS:
Mineral trioxide aggregate enhances the odonto/osteogenic capacity of DPSCs from inflammatory sites via activating the NF-κB pathway.
© 2013 John Wiley & Sons A/S. Published by John Wiley & Sons Ltd.
BACKGROUND:
Acute lung injury (ALI) is considered to be the major cause of respiratory failure in critically ill patients. Clinical studies have found that in patients with sepsis and after hemorrhage, the elevated level of high mobility group box-1(HMGB-1) in their circulation is highly associated with ALI, but the underlying mechanism remains unclear. Extracellular HMGB-1 has cytokine-like properties and can bind to Toll-like Receptor-4 (TLR4), which was reported to play an important role in the pathogenesis of ALI. The aim of this study was to determine whether HMGB-1 directly contributes to ALI and whether TLR4 signaling pathway is involved in this process.
METHODS:
Recombinant human HMGB-1 (rhHMGB-1) was used to induce ALI in male Sprague-Dawley rats. Lung specimens were collected 2 h after HMGB-1 treatment. The levels of TNF-α, IL-1β, TLR4 protein, and TLR4 mRNA in lungs as well as pathological changes of lung tissue were assessed. In cell studies, the alveolar macrophage cell line, NR8383, was collected 24 h after rhHMGB-1 treatment and the levels of TNF-α and IL-1β in cultured medium as well as TLR4 protein and mRNA levels in the cell were examined. TLR4-shRNA-lentivirus was used to inhibit TLR4 expression, and a neutralizing anti-HMGB1 antibody was used to neutralize rhHMGB-1 both in vitro and in vivo.
RESULTS:
Features of lung injury and significant elevation of IL-1β and TNF-α levels were found in lungs of rhHMGB-1-treated animals. Cultured NR8383 cells were activated by rhHMGB-1 treatment and resulted in the release of IL-1β and TNF-α. TLR4 expression was greatly up-regulated by rhHMGB-1. Inhibition of TLR4 or neutralization of HMGB1 with a specific antibody also attenuated the inflammatory response induced by HMGB-1 both in vivo and in vitro.
CONCLUSION:
HMGB-1 can activate alveolar macrophages to produce proinflammatory cytokines and induce ALI through a mechanism that relies on TLR-4.
Use of a skin flap has been a common technique in reconstructive surgery for more than five decades. However, partial necrosis of its distal end is still a serious postoperative complication. Many theories about this problem have been proposed, including deficient blood supply, which is the most accepted theory. In this study we demonstrated that hypoxic preconditioning enhanced the viability of adipose-derived stem cells (ADSCs) in vivo and improved their ability to increase the survival rate of ischemic skin flaps in rats. Seven days after flap elevation, the flap survival rate in the hypoxic preconditioned ADSC group was higher than that in the control group. Moreover, histological examination showed that more ADSCs survived in flaps treated by hypoxic preconditioning. Vascular density in the hypoxic preconditioned ADSC group was 30-90 % greater than that in the control group. In addition, the expressions of vascular endothelial growth factor and hypoxia inducible factor-1α (HIF-1α) were higher in the hypoxic preconditioned ADSC group than in the control group (p < 0.05). This enhancive phenomenon reached its highest level at the precondition times of 3 and 7 days in the hypoxic preconditioned ADSC group. We conclude that hypoxia preconditioning effectively enhances the viability of ADSCs to increase the survival rate of ischemic skin flaps. Furthermore, 3 days is the optimal preconditioning time point.
LEVEL OF EVIDENCE II:
This journal requires that authors assign a level of evidence to each article. For a full description of these Evidence-Based Medicine ratings, please refer to the Table of Contents or the online Instructions to Authors www.springer.com/00266 .
AIM:
This study was performed to address the bone injury and the early molecular responses of bone to obstructive nephropathy induced by unilateral ureteral obstruction in mice.
METHODS:
The male mice were subjected to unilateral ureteral obstruction (UUO, n = 10) or sham operation (n = 10). All mice were killed on day 7 after the surgical operation. Hematoxylin and eosin and tartate-resistant acid phosphatase staining were performed on paraffin-embedded bone sections. Expression of genes and proteins was analyzed by reverse transcription-polymerase chain reaction, and Western blotting and immunohistochemistry staining, respectively.
RESULTS:
The serum calcium level was significantly reduced in UUO mice compared with that of Sham mice. The proximal tibia of UUO mice exhibited the increased expansion of chondrocytes zone, the reduction of osteoid content, and the increased separation and disconnection of woven bones. Reverse transcription-polymerase chain reaction results showed the downregulation of Cbfa1 and Col mRNA expression and the upregulation of Tgf-β, CtsK, CaII, Opg and Rankl mRNA expression in tibia of UUO mice compared to those of Sham mice. The ratio of Opg and Rankl was unchanged between Sham and the UUO group. Local protein expression of angiotensin II and its type 2 receptor was dramatically upregulated in tibia of UUO mice.
CONCLUSION:
Together, it is concluded that the obstructive nephropathy has defective effects on bone, and the underlying mechanisms are the reduction of bone formation and the increase of bone resorption, which is mediated, at least partially through local angiotensin II signalling.
© 2012 The Authors. Nephrology © 2012 Asian Pacific Society of Nephrology.