Unconjugated
Lipoxin A4 (LXA4), an endogenous arachidonic acid metabolite, was previously considered an anti-inflammatory lipid mediator. But it also has the potential to inhibit cancer progression. To explore the therapeutic effect of LXA4 in pancreatic cancer, we used Panc-1 cells to investigate the mechanism by which LXA4 can attenuate pancreatic cancer cell invasion. Our data showed that LXA4 significantly inhibited both cell invasion and the expression of matrix metalloproteinase- (MMP-) 9 and MMP-2. Further experiments implied that LXA4 decreased the levels of intracellular reactive oxygen species (ROS) and the activity of the extracellular signal regulated kinases (ERK) pathway to achieve similar outcome to ROS scavenger N-acetyl-L-cysteine (NAC). However, a decreased level of intracellular ROS was not observed in cells treated with the specific ERK pathway inhibitor FR180204. The blocking of either intracellular ROS or ERK pathway caused the downregulation of MMP-9 and MMP-2 expression. Furthermore, tests revealed that LXA4 inhibited MMP-9 and MMP-2 at the mRNA, protein, and functional levels. Finally, LXA4 dramatically limited the invasion of CoCl2-mimic hypoxic cells and abrogated intracellular ROS levels, ERK activity, and MMPs expression. These results suggest that LXA4 attenuates cell invasion in pancreatic cancer by suppressing the ROS/ERK/MMPs pathway, which may be beneficial for preventing the invasion of pancreatic cancer.
Bone marrow stroma plays a critical role in the bone metastasis of breast cancer. Bone marrow-derived mesenchymal stem cells (BMSC) are critical to facilitate cancer progression. Human bone morphogenetic protein 9 (BMP9) is the most potent osteogenic factor and one of bone-stored growth factors involved in both promotion and inhibition of different cancers. However, it is unclear whether BMP9 correlates with the bone metastasis of breast cancer. This study was to evaluate the role of BMP9 in the interaction between BMSC and breast cancer cells (BCC). To determine whether BMP9 is able to block the tumor promoting effect of BMSC, an in vitro model was developed using breast cancer MDA-MB-231 cells co-cultured with bone marrow-derived mesenchymal stem cells HS-5 with-BMP9 overexpression. The expressions of metastasis-related genes were detected to identify important factors mediating the role of BMP9 in breast cancer cells. Results showed BMP9 could inhibit invasion and promote apoptosis of MDA-MB-231 cells. The expressions of interleukin-6 (IL-6), matrix metalloproteinase-2 (MMP-2) and monocyte chemoattratctant protein-1 (MCP-1) decreased in the MDA-MB-231 cells of BMP9 over-expression group, and the expressions of epithelial-mesenchymal transition (EMT)-related molecules was also reduced. On the other hand, the expression of stromal cell derived factor-1 (SDF-1) decreased in HS-5 cells of BMP9 over-expression group. Taken together, BMP9 is able to inhibit the migration and promote the apoptosis of breast cancer by regulating the interaction between MDA-MB-231 cells and HS-5 cells in which SDF-1/CXCR4-PI3K pathway and EMT are involved.
Naked cuticle homolog2 (NKD2) is located in chromosome 5p15.3, which is frequently loss of heterozygosity in human colorectal and gastric cancers. In order to understand the mechanism of NKD2 in gastric cancer development, 6 gastric cancer cell lines and 196 cases of human primary gastric cancer samples were involved. Methylation specific PCR (MSP), gene expression array, flow cytometry, transwell assay and xenograft mice model were employed in this study. The expression of NKD1 and NKD2 was silenced by promoter region hypermethylation. NKD1 and NKD2 were methylated in 11.7% (23/196) and 53.1% (104/196) in human primary gastric cancer samples. NKD2 methylation is associated with cell differentiation, TNM stage and distant metastasis significantly (all P < 0.05), and the overall survival time is longer in NKD2 unmethylated group compared to NKD2 methylated group (P < 0.05). Restoration of NKD2 expression suppressed cell proliferation, colony formation, cell invasion and migration, induced G2/M phase arrest, and sensitized cancer cells to docetaxel. NKD2 inhibits SOX18 and MMP-2,7,9 expression and suppresses BGC823 cell xenograft growth. In conclusion, NKD2 methylation may serve as a poor prognostic and chemo-sensitive marker in human gastric cancer. NKD2 impedes gastric cancer metastasis by inhibiting SOX18.
Fangchinoline, an important compound in Stephania tetrandra S. Moore, as a novel antitumor agent, has been implicated in several types of cancers cells except gastric cancer. To investigate whether fangchinoline affects gastric cancer cells, we detected the signaling pathway by which fangchinoline plays a role in different human gastric cancer cells lines. We found that fangchinoline effectively suppressed proliferation and invasion of SGC7901 cell lines, but not MKN45 cell lines by inhibiting the expression of PI3K and its downstream pathway. All of the Akt/MMP2/MMP9 pathway, Akt/Bad pathway, and Akt/Gsk3β/CDK2 pathway could be inhibited by fangchinoline through inhibition of PI3K. Taken together, these results suggest that fangchinoline targets PI3K in tumor cells that express PI3K abundantly and inhibits the growth and invasive ability of the tumor cells.
Increased levels of angiotensin II (Ang II) and activated matrix metalloproteinase 2 (MMP-2) produced by human aortic smooth muscle cells (human ASMCs) have recently been implicated in the pathogenesis of thoracic aortic aneurysm (TAA). Additionally, angiotensin II type 1 receptor (AT1R)-mediated extracellular signal-regulated kinase (ERK)1/2 activation contributes to TAA development in Marfan Syndrome. However, there is scant data regarding the relationship between Ang II and MMP-2 expression in human ASMCs. Therefore, we investigated the effect of Ang II on MMP-2 expression in human ASMCs and used Western blotting to identify the Ang II receptors and intracellular signaling pathways involved. Reverse transcription polymerase chain reaction (RT-PCR) and immunofluorescence data demonstrated that Ang II receptors were expressed on human ASMCs. Additionally, Ang II increased the expression of Ang II type 2 receptor (AT2R) but not AT1R at both the transcriptional and translational levels. Furthermore, Western blotting showed that Ang II increased MMP-2 expression in human ASMCs in a dose- and time-dependent manner. This response was completely inhibited by the AT1R inhibitor candesartan but not by the AT2R blocker PD123319. In addition, Ang II-induced upregulation of MMP-2 was mediated by the activation of ERK1/2, whereas p38 mitogen-activated protein kinase (p38 MAPK) and c-Jun N-terminal kinase (JNK) had no effect on this process. In conclusion, these results indicate that Ang II can increase the expression of MMP-2 via AT1 receptor and ERK1/2 signaling pathways in human ASMCs and suggest that antagonists of AT1R and ERK1/2 may be useful for treating TAAs.
Perineural invasion (PNI) is considered as an alternative route for the metastatic spread of pancreatic cancer cells; however, the molecular changes leading to PNI are still poorly understood. In this study, we show that the CXCL12/CXCR4 axis plays a pivotal role in the neurotropism of pancreatic cancer cells to local peripheral nerves. Immunohistochemical staining results revealed that CXCR4 elevation correlated with PNI in 78 pancreatic cancer samples. Both in vitro and in vivo PNI models were applied to investigate the function of the CXCL12/CXCR4 signaling in PNI progression and pathogenesis. The results showed that the activation of the CXCL12/CXCR4 axis significantly increased pancreatic cancer cells invasion and promoted the outgrowth of the dorsal root ganglia. CXCL12 derived from the peripheral nerves stimulated the invasion and chemotactic migration of CXCR4-positive cancer cells in a paracrine manner, eventually leading to PNI. In vivo analyses revealed that the abrogation of the activated signaling inhibited tumor growth and invasion of the sciatic nerve toward the spinal cord. These data indicate that the CXCL12/CXCR4 axis may be a novel therapeutic target to prevent the perineural dissemination of pancreatic cancer.
Dapper, Dishevelled-associated antagonist of β-catenin (DACT), is a key regulator of Wnt signaling pathway. The purpose of this study is to explore the epigenetic changes and the function ofDACT2 in human gastric cancer (GC). Eight human gastric cancer cell lines, 167 cases of primary gastric cancer and 8 cases of normal gastric mucosa were involved in this study. In addition, methylation Specific PCR (MSP), semi-quantitative RT-PCR, colony formation assay, flow cytometry assay, siRNA, immunofluorescence techniques and xenograft mice models were employed. The results indicate that DACT2 is frequently methylated in human primary gastric cancer (55.7%), and that methylation of DACT2 is associated with lost or reduction in its expression (X(2) test, P<0.01). We found that DACT2 expression was regulated by promoter region hypermethylation. Methylation of DACT2 is associated with tumor differentiation, invasion and intravascular cancerous emboli (X(2) test, P<0.05, P<0.05 and P<0.05). In gastric cancer patients treated with 5-FU and cisplatin, the five-year survival rates are higher in DACT2 methylated cases. DACT2 inhibits cell proliferation, migration and invasion in gastric cancer cells and suppresses gastric cancer xenografts in mice. Restoration of DACT2 expression inhibits both canonical and noncanonical WNT signaling in SGC7901 cells. Restoration of DACT2 expression sensitized gastric cancer cells to paclitaxel and 5-FU. In conclusion, DACT2 is frequently methylated in human gastric cancer and DACT2 expression is silenced by promoter region hypermethylation. DACT2 suppressed gastric cancer proliferation, invasion and metastasis by inhibiting Wnt signaling both in vitro and in vivo.
The study investigated the effect of high- and low-intensity exercise training on inflammatory reaction of blood and skeletal muscle in streptozotocin (STZ)-induced diabetic male Sprague-Dawley rats (243 ± 7 g, 8 weeks). The rats completed treadmill running in either high-intensity exercise (6 weeks of exercise training, acute bouts of exercise) or low-intensity exercise (6 weeks of exercise training). Non-running, sedentary rats served as controls. To induce diabetes mellitus, rats received a peritoneal injection of STZ (50 mg · kg(-1)). Rats were sacrificed immediately after an acute bout of exercise and 6 weeks of exercise training. Inflammatory factors were analyzed by ELISA and by immune blotting from the soleus and extensor digitorum longus muscles. In the serum, inflammatory cytokines (IL-1β, TNF-α, IL-6, IL-4) and reactive oxygen species (ROS) (nitric oxide and malondialdehyde) increased in diabetic rats. However, all exercise training groups displayed reduced inflammatory cytokines and reactive oxygen species. In skeletal muscles, low-intensity exercise training, but not high intensity exercise, reduced the levels of COX-2, iNOS, and MMP-2, which were otherwise markedly elevated in the presence of STZ. Moreover, the levels of GLUT-4 and MyoD were effectively increased by different exercise intensity and exercise duration. Low-intensity exercise training appeared most effective to reduce diabetes-related inflammation. However, high-intensity training also reduced inflammatory factors in tissue-specific muscles. The data implicate regular exercise in protecting against chronic inflammatory diseases, such as diabetes.
Gastric cancer (GC) is the fourth most common malignancy in males and the fifth most common malignancy in females worldwide. DACH1 is frequently methylated in hepatic and colorectal cancer. To further understand the regulation and mechanism of DACH1 in GC, eight GC cell lines, eight cases of normal gastric mucosa, 98 cases of primary GC and 50 cases of adjacent non-tumour tissues were examined. Methylation-specific PCR, western blot, transwell assay and xenograft mice were used in this study. Loss of DACH1 expression correlated with promoter region methylation in GC cells, and re-expression was induced by 5-Aza-2'-deoxyazacytidine. DACH1 is methylated in 63.3% (62/98) of primary GC and 38% (19/50) of adjacent non-tumour tissues, while no methylation was found in normal gastric mucosa. Methylation of DACH1 correlated with reduced expression of DACH1 (P < 0.01), late tumour stage (stage III/IV) (P < 0.01) and lymph node metastasis (P < 0.05). DACH1 expression inhibited epithelial-mesenchymal transition and metastasis by inhibiting transforming growth factor (TGF)-β signalling and suppressed GC cell proliferation through inducing G2/M phase arrest. The tumour size is smaller in DACH1-expressed BGC823 cell xenograft mice than in unexpressed group (P < 0.01). Restoration of DACH1 expression also sensitized GC cells to docetaxel. These studies suggest that DACH1 is frequently methylated in human GC and expression of DACH1 was controlled by promoter region methylation. DACH1 suppresses GC proliferation, invasion and metastasis by inhibiting TGF-β signalling pathways both in vitro and in vivo. Epigenetic silencing DACH1 may induce GC cells' resistance to docetaxel.
Cathepsin B (CB), an important proteinase that participates in joint destruction in rheumatoid arthritis (RA), exhibits higher expression in fibroblast-like synoviocyte (FLS) of abnormal proliferative synovial tissues. Whether and how it affects the biological behaviours of RA-FLS, such as migration and invasion, are poorly understood. In the present study, CB expression in synovial tissues of patients with RA and ostearthritis (OA) were measured by quantitative polymerase chain reaction (qPCR) and immunohistochemistry (IHC), respectively. Stable depletion of endogenous CB was achieved by small interfering RNA (siRNA) transfection, and decrease of CB activity was acquired by using its specific inhibitor (CA074Me). The effects of CA074Me and RNA interference (RNAi) treatments on proliferation, migration, invasion, matrix metalloproteinase (MMP)-2/-9 expression, focal adhesion kinase (FAK) activation, and mitogen-activated protein kinases (MAPKs) phosphorylation of FLS were analysed. In RA synovial tissues, CB was expressed at elevated levels compared with OA synovial tissues. CA074Me could inhibit invasion of FLS obtained from RA patients in an ex-vivo invasion model. CA074Me and siRNA treatments suppressed the migration and invasion of FLS, reduced the activity, expression and mRNA level of MMP-2, restrained the activation of FAK and reduced the expression of F-actin. Moreover, CA074Me decreased the phosphorylation of P38 MAPK and c-Jun N-terminal kinase (JNK) in FLS, while siCB treatment reduced the phosphorylation of P38 but not JNK. CB substantially contributes to the invasive phenotype of FLS that leads to joint destruction in RA. This proteinase may show promise as a therapeutic target in inflammatory arthritis.
Granulocyte/macrophage colony-stimulating factor (GM-CSF) can accelerate wound healing by promoting angiogenesis. The biological effects of GM-CSF in angiogenesis and the corresponding underlying molecular mechanisms, including in the early stages of primitive endothelial tubule formation and the later stages of new vessel maturation, have only been partially clarified. This study aimed to investigate the effects of GM-CSF on angiogenesis and its regulatory mechanisms. Employing a self-controlled model (Sprague-Dawley rats with deep partial-thickness burn wounds), we determined that GM-CSF can increase VEGF expression and decrease the expression ratio of Ang-1/Ang-2 and the phosphorylation of Tie-2 in the early stages of the wound healing process, which promotes the degradation of the basement membrane and the proliferation of endothelial cells. At later stages of wound healing, GM-CSF can increase the expression ratio of Ang-1/Ang-2 and the phosphorylation of Tie-2 and maintain a high VEGF expression level. Consequently, pericyte coverages were higher, and the basement membrane became more integrated in new blood vessels, which enhanced the barrier function of blood vessels. In summary, we report here that increased angiogenesis is associated with GM-CSF treatment, and we indicate that VEGF and the Ang/Tie system may act as angiogenic mediators of the healing effect of GM-CSF on burn wounds.
To explore epigenetic regulation and the impact of chemokine CXCL14 on colorectal cancer, 7 colorectal cancer cell lines, 107 cases of primary colorectal cancer, and 10 cases of normal colorectal mucosa were evaluated in this study. Methylation specific PCR (MSP), semi-quantitative reverse-transcription PCR (RT-PCR), cell proliferation assay, colony formation, and transwell assay were performed for the evaluation. Complete methylation and loss of CXCL14 expression were found in 5 colorectal cancer cell lines. Partial methylation and weak expression were found in two cell lines. CXCL14 was methylated in 79.4% (85/107) of primary human colorectal cancer. No methylation was found in 10 cases of normal colorectal mucosa. Restoration of CXCL14 expression was induced by the 5-aza-2'-deoxycytidine (DAC) treatment. The cell viability was reduced and colony formation was inhibited by restoration of CXCL14 expression in HCT116 cells, a colorectal cancer cell line. The number of invasive and migration cells was reduced by CXCL14. The expression of MMP-2, Vimentin, and NF-κB was suppressed, and the expression of E-cadherin and IκB-α was induced by CXCL14. In conclusion, CXCL14 is frequently methylated in human colorectal cancer and promoter region hypermethylation silenced CXCL14 expression in colorectal cancer cells. Restoration of CXCL14 expression suppressed colorectal cancer proliferation. CXCL14 inhibits colorectal cancer migration, invasion, and epithelial-to-mesenchymal transition (EMT) by suppressing NF-κB signaling.
Aberrant expression of histone deacetylases (HDACs) is associated with carcinogenesis. Some HDAC inhibitors are widely considered as promising anticancer therapeutics. A major obstacle for development of HDAC inhibitors as highly safe and effective anticancer therapeutics is that our current knowledge on the contributions of different HDACs in various cancer types remains scant. Here we report that the expression level of HDAC10 was significantly lower in patients exhibiting lymph node metastasis compared with that in patients lacking lymph node metastasis in human cervical squamous cell carcinoma. Forced expression of HDAC10 in cervical cancer cells significantly inhibited cell motility and invasiveness in vitro and metastasis in vivo. Mechanistically, HDAC10 suppresses expression of matrix metalloproteinase (MMP) 2 and 9 genes, which are known to be critical for cancer cell invasion and metastasis. At the molecular level, HDAC10 binds to MMP2 and -9 promoter regions, reduces the histone acetylation level, and inhibits the binding of RNA polymerase II to these regions. Furthermore, an HDAC10 mutant lacking histone deacetylase activity failed to mimic the functions of full-length protein. These results identify a critical role of HDAC10 in suppression of cervical cancer metastasis, underscoring the importance of developing isoform-specific HDAC inhibitors for treatment of certain cancer types such as cervical squamous cell carcinoma.
MicroRNAs (miRs) are known to have an important role in modulating vascular biology. MiR21 was found to be involved in the pathogenesis of proliferative vascular disease. The role of miR21 in endothelial cells (ECs) has well studied in vitro, but the study in vivo remains to be elucidated. In this study, miR21 endothelial-specific knockout mice were generated by Cre/LoxP system. Compared with wild-type mice, the miR21 deletion in ECs resulted in structural and functional remodeling of aorta significantly, such as diastolic pressure dropping, maximal tension depression, endothelium-dependent relaxation impairment, an increase of opening angles and wall-thickness/inner diameter ratio, and compliance decrease, in the miR21 endothelial-specific knockout mice. Furthermore, the miR21 deletion in ECs induced down-regulation of collagen I, collagen III and elastin mRNA and proteins, as well as up-regulation of Smad7 and down-regulation of Smad2/5 in the aorta of miR21 endothelial-specific knockout mice. CTGF and downstream MMP/TIMP changes were also identified to mediate vascular remodeling. The results showed that miR21 is identified as a critical molecule to modulate vascular remodeling, which will help to understand the role of miR21 in vascular biology and the pathogenesis of vascular diseases.
AIMS:
To study the proliferation of osteoblasts and genes expression under normal glucose, high glucose, and metformin (Met).
METHODS:
MG63 osteoblast-like cells were cultured in osteogenic medium supplemented with normal glucose (glucose 5.5 mmol/L) or high glucose (glucose 16.7 mmol/L) and metformin + high glucose (Met 300 μmol/L + glucose 16.7 mmol/L). Proliferation was detected with CCK-8 assay at days 1, 3, and 7. Real-time PCR and Western blot were performed to compare the expression of collagen I (Col I), osteocalcin (OCN), osteoprotegerin (OPG), receptor activator for NF- κB ligand (RANKL), and metal matrix proteinases 1 and 2 (MMP1, MMP2). Alkaline phosphatase (ALP) activity was also detected at days 6, 12, and 18.
RESULTS:
Exposure to high glucose inhibited the proliferation of osteoblasts (P < 0.05), with suppressed OCN and OPG. Meanwhile, Col I, RANKL, MMP1, and MMP2 were unaffected. Metformin attenuated the suppression on proliferation with increased expression of Col I, OCN, and OPG, meanwhile suppressing MMP1 and MMP2. High glucose lowered the intracellular ALP, while metformin raised it. Metformin attenuated the downregulation of ALP completely at day 6, partly at day 12, but not at day 18.
CONCLUSIONS:
Metformin attenuated the suppression effect of high glucose to the osteoblast proliferation and gene expression, more prominently in earlier stage.
AIM:
To explore whether icaritin, a prenylflavonoid derivative of the Chinese tonic herb Epimedium, could suppress the proliferation of human osteosarcoma cells in vitro, and to elucidate the mechanisms of the action.
METHODS:
Human osteosarcoma SaOS2 cell line was used in the present study. The proliferation of the cells was examined using MTT assay and immunofluorescence DAPI staining. Cell motility was studied with the scratch assay. Cell apoptosis was determined by Annexin V-FITC and PI double staining using flow cytometry. Western blotting and RT-PCR were used to measure the expression of mRNAs and proteins in the cells.
RESULTS:
Icaritin (5-15 μmol/L) suppressed the proliferation of SaOS2 cells in vitro in a dose-dependent manner. Furthermore, the cell motility was significantly decreased after exposure to icaritin. Moreover, icaritin (5 μmol/L) time-dependently induced the apoptosis of SaOS2 cells, markedly suppressed MMP-2 and MMP-9 expression, upregulated caspase-3 and caspase-9 expression, and increased the level of cleaved caspase-3 in the cells. Co-exposure to the caspase-3 inhibitor zVAD-fmk (10 μmol/L) compromised the icaritin-induced caspase-3 expression and apoptosis in SaOS2 cells.
CONCLUSION:
Icaritin suppresses the proliferation of SaOS2 human osteosarcoma cells by increasing apoptosis and downregulating MMP expression.
BACKGROUND:
CDK11p58 is one of the large families of p34cdc2-related kinases whose functions are linked with cell cycle progression, tumorigenesis and apoptotic signaling. Our previous investigation demonstrated that CDK11p58 repressed androgen receptor (AR) transcriptional activity and was involved in the negative regulation of AR function.
METHODS:
CDK11p58 expression was examined in the prostate cancer tissues and adjacent tissues by IHC and qRT-PCR. Cell apoptosis was detected by flow cytometry. The metastasis of cancer cells was evaluated by the Transwell Assay. Finally we further investigated the underlying molecular mechanisms by examining expression levels of relevant proteins using western blot analysis.
RESULTS:
We found that both RNA and protein expression of CDK11p58 were low in prostate cancer tissues compared with its adjacent noncancerous tissues. CDK11p58 promoted the prostate cancer cell apoptosis and inhibited its metastasis in a kinase dependent way. And finally CDK11p58 could inhibit the metastasis of AR positive prostate cancer cells through inhibition of integrin β3 and MMP2.
CONCLUSIONS:
These data indicate that CDK11p58 is an anti-metastasis gene product in prostate cancer.
BACKGROUND:
Growing evidence suggests that miR-29a has an important role in regulating tumourigenesis and development of various types of cancer. However, the role and the underlying mechanism of miR-29a in colorectal cancer (CRC) remain largely unknown.
METHODS:
MiR-29a targeted gene was identified by the luciferase assay and western blot. MiR-29a function was analysed by invasion assays and the orthotopic transplantation mouse model. The miR-29a pathway was assayed by real-time PCR, western blot and chip analysis.
RESULTS:
KLF4 was identified as a direct target gene of miR-29a. MiR-29a promoted CRC cell invasion, which was blocked by re-expression of KLF4. In addition, MMP2 was identified as a novel direct target of KLF4. Both miR-29a overexpression and KLF4 knockdown promoted MMP2 expression but inhibited E-cadherin expression. Furthermore, clinical data indicated that both miR-29a high expression and KLF4 mRNA low expression were associated with metastasis and poor prognosis in CRC patients, and KLF4 protein expression was inversely correlated with MMP2 but positively correlated with E-cad protein expression.
CONCLUSION:
Increased expression of miR-29a promoted CRC metastasis by regulating MMP2/E-cad through direct targeting KLF4, which highlights the potential of the miR-29a inhibitor as a novel agent against CRC metastasis.
BACKGROUND:
Matrix metalloproteinase 2 (MMP-2) has been reported to be an important regulator of cell migration and invasion through degradation of the extracellular matrix (ECM) in many diseases, such as cancer and endometriosis. Here, we found calcium-activated neutral protease 7 (CAPN 7) expression was markedly upregulated in the eutopic endometrium and endometrial stromal cells of women diagnosed with endometriosis. Our studies were carried out to detect the effects of CAPN 7 on human endometrial stromal cell (hESC) migration and invasion.
METHODS:
Western blotting and quantitative real-time PCR were used to detect the expression of CAPN 7 in endometriosis patients and normal fertile women. Scratch-wound-healing and invasion chamber assay were used to investigate the role of CAPN 7 in hESC migration and invasion. Western blotting, quantitative real-time PCR and zymography were carried out to detect the effect of CAPN 7 on the expressions and activity of MMP-2.
RESULTS:
CAPN 7 was markedly up-regulated in endometriosis, thereby promoting the migration and invasion of hESC. CAPN 7 overexpression led to increased expression of MMP-2 and tissue inhibitor of metalloproteinases 2 (TIMP-2); CAPN 7 knockdown reversed these changes. CAPN 7 increased MMP-2 activity by increasing the ratio of MMP-2 to TIMP-2. We also found that OA-Hy (an MMP-2 inhibitor) decreased the effects of CAPN 7 overexpression on hESC migration and invasion by approximately 50% and 55%, respectively. Additionally, a coimmunoprecipitation assay demonstrated that CAPN 7 interacted with activator protein 2α (AP-2α): an important transcription factor of MMP-2.
CONCLUSIONS:
CAPN 7 promotes hESC migration and invasion by increasing the activity of MMP-2 via an increased ratio of MMP-2 to TIMP-2.
AIM:
To evaluate the protective role of AE-941, a matrix metalloproteinase (MMP) inhibitor, on ulcerative colitis (UC) in rats.
METHODS:
Sprague Dawley (SD) rats were randomly divided into three groups: a control group, an AE-941 treatment group, and an UC model group. Rats were sacrificed on days 7, 21, or 56 following administration of treatment by enema and the disease activity index (DAI), colonic mucosa damage index (CMDI) and colonic expression of MMP-2 and MMP-9 were assessed.
RESULTS:
DAI and CDMI scores in the UC model group increased significantly compared to the control group at all timepoints (P < 0.001), and also increased significantly at the 21- and 56-d timepoints compared to the AE-941-treated group (DAI: 21- and 56-d = 2.09 ± 0.25, 1.52 ± 0.30 vs 1.55 ± 0.28, 0.59 ± 0.19, respectively, P = 0.040 and 0.007, CMDI: 21- and 56-d = 3.03 ± 0.42, 1.60 ± 0.35 vs 2.08 ± 0.46, 0.86 ± 0.37, respectively, P = 0.040 and 0.005). Furthermore, the colonic expression of MMP-2 and MMP-9 in the UC model group increased significantly compared to the control group (P < 0.001), and also increased compared to the AE-941-treated group on the 21- and 56-d timepoints (MMP-2: 21- and 56-d = 0.6048 ± 0.0522, 0.4163 ± 0.0330 vs 0.3983 ± 0.0218, 0.1093 ± 0.0072, respectively, P = 0.010; MMP-9: 21- and 56-d = 0.6873 ± 0.0472, 0.4328 ± 0.0257 vs 0.5179 ± 0.0305, 0.2673 ± 0.0210, respectively, P = 0.010 and 0.040).
CONCLUSION:
Expression of MMP-2 and MMP-9 increased significantly in rats with UC. AE-941 can reduce colonic mucosal damage by downregulating the expression of MMP-2 and MMP-9.
AIMS:
To explore the epigenetic changes and the function of TFPI-2 in esophageal cancer.
MATERIALS & METHODS:
Nine esophageal cancer cell lines, nine normal esophageal mucosa, 60 esophageal dysplasia and 106 advanced esophageal cancer samples were included in this study. TFPI-2 methylation was examined by methylation-specific PCR. TFPI-2 expression was evaluated by immunohistochemistry in tissue samples. The effect of TFPI-2 on proliferation, apoptosis, invasion and migration was analyzed by colony formation assay, western blot assay, transwell assay and flow cytometric analysis.
RESULTS:
TFPI-2 expression was regulated by promoter region hypermethylation in human esophageal cancer cell lines, and TFPI-2 expression is inversely correlated with methylation in primary cancer. Methylation was found in 28.2, 33.3 and 33.3% of grade 1, 2 and 3 esophageal dysplasia, and 67% of primary esophageal cancer, but no methylation was found in normal mucosa. Methylation is significantly related to tumor differentiation. Inhibition of invasion, migration, colony formation and proliferation, and induction of apoptosis occurred with the restoration of TFPI-2 expression in the KYSE70 cell line.
CONCLUSION:
TFPI-2 is frequently methylated in esophageal cancer with a progression tendency. TFPI-2 is a potential tumor suppressor in esophageal cancer.
Neotuberostemonine (NTS) is one of the main antitussive alkaloids in the root of Stemona tuberosa Lour. This study aimed to investigate the effects of NTS on bleomycin (BLM)-induced pulmonary fibrosis in mice and the underlying mechanism. After BLM administration, NTS were orally administered to mice at 20 and 40mg/kg per day from days 8 to 21, with nintedanib as a positive control. The effect of NTS on BLM-induced mice was assessed via histopathological examination by HE and Masson's trichrome staining, TGF-β1 level and macrophage recruitment by immunohistochemical staining, expression of profibrotic media and M1/M2 polarization by western blot. RAW 264.7 cells were used to evaluate whether NTS (1, 10, 100μM) directly affected macrophages. The results revealed that NTS treatment significantly ameliorated lung histopathological changes and decreased inflammatory cell counts in the bronchoalveolar lavage fluid. The over-expression of collagen, α-SMA and TGF-β1 was reduced by NTS. Furthermore, NTS markedly lowered the expression of MMP-2 and TIMP-1 while raised the expression of MMP-9. A further analysis showed that NTS was able to decrease the recruitment of macrophages and to inhibit the M2 polarization in mice lung tissues. The experiment in vitro showed that NTS significantly reduced the arginase-1 (marker for M2) expression in a dose-dependent manner but down-regulated the iNOS (marker for M1) expression only at 100μM. In conclusion, our study demonstrated for the first time that NTS has a significant protective effect on BLM-induced pulmonary fibrosis through suppressing the recruitment and M2 polarization of macrophages.
EPB41L3 may play a role as a metastasis suppressor by supporting regular arrangements of actin stress fibres and alleviating the increase in cell motility associated with enhanced metastatic potential. Downregulation of epb41l3 has been observed in many cancers, but the role of this gene in esophageal squamous cell carcinoma (ESCC) remains unclear. Our study aimed to determine the effect of epb41l3 on ESCC cell migration and invasion. We investigated epb41l3 protein expression in tumour and non-tumour tissues by immunohistochemical staining. Expression in the non-neoplastic human esophageal cell line Het-1a and four ESCC cell lines - Kyse150, Kyse510, Kyse450 and Caes17 - was assessed by quantitative Polymerase Chain Reaction (qPCR) and Western blotting. Furthermore, an EPB41L3 overexpression plasmid and EPB41L3-specific small interfering RNA were used to upregulate EPB41L3 expression in Kyse150 cells and to downregulate EPB41L3 expression in Kyse450 cells, respectively. Cell migration and invasion were evaluated by wound healing and transwell assays, respectively. The expression levels of p-AKT, matrix metalloproteinase (MMP)2 and MMP9 were evaluated. Expression of epb41l3 was significantly lower in tumour tissues than in non-tumour tissues and in ESCC cell lines compared with the Het-1a cell line. Kyse450 and Caes17 cells exhibited higher expression of epb41l3 than Kyse150 and Kyse510 cells. Overexpressing epb41l3 decreased Kyse150 cell migration and invasion, whereas EPB41L3-specific small interfering RNA silencing increased these functions in Kyse450 cells. Furthermore, overexpressing epb41l3 led to downregulation of MMP2 and MMP9 in Kyse150 and Kyse510 cells. Our findings reveal that EPB41L3 suppresses tumour cell invasion and inhibits MMP2 and MMP9 expression in ESCC cells.
(-)-β-hydrastine is one of the main active components of the medicinal plant, Hydrastis canadensis, which is used in many dietary supplements intended to enhance the immune system. However, whether (-)-β-hydrastine affects the tumor signaling pathway remains unexplored. In the present study, we found that (-)-β-hydrastine inhibited the kinase activity of p21-activated kinase 4 (PAK4), which is involved in the regulation of cytoskeletal reorganization, cell proliferation, gene transcription, oncogenic transformation and cell invasion. In the present study, (-)-β-hydrastine suppressed lung adenocarcinoma cell proliferation by inhibiting expression of cyclin D1/D3 and CDK2/4/6, leading to cell cycle arrest at the G1 phase, in a PAK4 kinase-dependent manner. Moreover, inhibition of PAK4 kinase activity by (-)-β-hydrastine also promoted the early apoptosis of lung adenocarcinoma cells through the mitochondrial apoptosis pathway. In addition, (-)-β-hydrastine significantly suppressed the migration and invasion of human lung adenocarcinoma cells in conjunction with concomitant blockage of the PAK4/LIMK1/cofilin, PAK4/SCG10 and PAK4/MMP2 pathways. All of these data indicate that (-)-β-hydrastine, as a novel PAK4 inhibitor, suppresses the proliferation and invasion of lung adenocarcinoma cells. Taken together, these results provide novel insight into the development of a PAK4 kinase inhibitor and a potential therapeutic strategy for lung cancer.
Dp71 is one of the most ubiquitously expressed isoforms of dystrophin, the pathological genes of DMD. In order to find whether the alteration of Dp71 can affect the phenotypes of cell other than PC12, an A549 cell line with stably transfected Dp71 siRNA plasmids was set up and named A549-Dp71AS cell. It is demonstrated for the first time that the A549-Dp71AS cell line displayed decreased invasion capabilities, reduced migration ability, decreased proliferation rate, and lessened clonogenic formation. Cisplatin-induced apoptosis was also increased in A549-Dp71AS cell line via enhancing the Caspase 3, Caspase 8, and Caspase 9 activities. Knocking down Dp71 expression can significantly inhibit the A549 xenograft tumor growth in nude mice. The A549-Dp71AS cells and xenograft tumor tissues displayed reduced lamin B1, Bcl-2, and MMP2 protein expression, which accounts for the reduced malignancy of A549-Dp71AS cells in vivo and in vitro.
Aging is characterized by the progressive decline in mass and function of the skeletal muscle along with increased susceptibility to inflammation, oxidative stress, and atrophy. In this study, we investigate the effect of intermittent bout and single bout exercise training on inflammatory molecules in young (3 months) and old (22 months) male Sprague-Dawley rats. The rats were divided into 6 groups. Young and old rats were randomly assigned for control and two exercise training groups, single bout (S type): 30 min/day, 5 days/week for 6 weeks and intermittent bout (I type): three times for 10 min/day, 5 days/week for 6 weeks respectively. The exercise training was carried out by a treadmill at a speed of 15m/min (young) or 10 m/min (old) with a slope of 5°. After 48 h of the final exercise bout, muscle samples were collected for biochemical assay. I type exercise training reduced the serum levels of inflammatory molecules such as interleukin-1β (IL-1β), tumor necrosis factor-alpha (TNF-α), interleukin-6 (IL-6), and malondialdehyde (MDA) in old rats. By contrast, interleukin-4 (IL-4) and superoxide dismutase (SOD) were elevated. Consequently in skeletal muscles, inducible nitric oxide synthase (iNOS) and cyclooxygenase-2 (COX-2) were decreased significantly in the old group of I type. However, the matrix metalloproteinase-2 (MMP-2) level had no positive effects. Also, phosphorylation of mammalian target of rapamycin (p-mTOR) and myogenic differentiation (MyoD) were increased markedly in S and I types of old rats. These results suggest that I type exercise training appears more effective to reduce age-associated inflammatory molecules, and may recommend in regulating against chronic complicated disease induced by aging.
Apelin is an adipokine that has a critical role in the development of atherosclerosis, which may offer potential for therapy. Because migration of vascular smooth muscle cells (VSMCs) is a key event in the development of atherosclerosis, understanding its effect on the atherosclerotic vasculature is needed. Here we investigated the effect of apelin on VSMC migration and the possible signaling mechanism. In cultured rat VSMCs, apelin dose- and time-dependently promoted VSMC migration. Apelin increased the phosphorylation of Akt, whereas LY294002, an inhibitor of phosphatidylinositol 3-kinase (PI3K), and an Akt1/2 kinase inhibitor blocked the apelin-induced VSMC migration. Apelin dose-dependently induced phosphorylation of Forkhead box O3a (FoxO3a) and promoted its translocation from the nucleus to cytoplasm, which were blocked by LY294002 and Akt1/2 kinase inhibitor. Furthermore, apelin increased matrix metalloproteinase 2 (MMP-2) expression and gelatinolytic activity. Overexpression of a constitutively active, phosphorylation-resistant mutant, TM-FoxO3a, in VSMCs abrogated the effect of apelin on MMP-2 expression and VSMC migration. ARP101, an inhibitor of MMP-2, suppressed apelin-induced VSMC migration. Moreover, the levels of apelin, phosphorylated Akt, FoxO3a, and MMP-2 were higher in human carotid-artery atherosclerotic plaque than in adjacent normal vessels. We demonstrate that PI3K/Akt/FoxO3a signaling may be involved in apelin inducing VSMC migration. Phosphorylation of FoxO3a plays a central role in mediating the apelin-induced MMP-2 activation and VSMC migration.
P-selectin-mediated tumor cell adhesion to platelets is a well-established stage in the process of tumor metastasis. Through computerized structural analysis, we found a marine-derived polysaccharide, holothurian glycosaminoglycan (hGAG), behaved as a ligand-competitive inhibitor of P-selectin, indicating its potential to disrupt the binding of P-selectin to cell surface receptor and activation of downstream regulators of tumor cell migration. Our experimental data demonstrated that hGAG significantly inhibited P-selectin-mediated adhesion of tumor cells to platelets and tumor cell migration in vitro and reduced subsequent pulmonary metastasis in vivo. Furthermore, abrogation of the P-selectin-mediated adhesion of tumor cells led to down-regulation of protein levels of integrins, FAK and MMP-2/9 in B16F10 cells, which is a crucial molecular mechanism of hGAG to inhibit tumor metastasis. In conclusion, hGAG has emerged as a novel anti-cancer agent via blocking P-selectin-mediated malignant events of tumor metastasis.
Breast cancer is the most common cause of death among women. KIF3C, a member of kinesin superfamily, functions as a motor protein involved in axonal transport in neuronal cells. To explore the expression, regulation and mechanism of KIF3C in breast cancer, 4 breast cancer cell lines and 93 cases of primary breast cancer and paired adjacent tissues were examined. Immunohistochemistry, Real Time Polymerase Chain Reaction (RT-PCR), Western blot, flow cytometry, short hairpin RNA (shRNA) interference, 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT), colony formation techniques and xenograft mice model were used. We found that KIF3C was over-expressed in breast cancer tissues and such high KIF3C expression was also associated with tumor recurrence and lymph node metastasis. Silencing of KIF3C by shRNA inhibited epithelial-mesenchymal transition and metastasis by inhibiting TGF-β signaling and suppressed breast cancer cell proliferation through inducing G2/M phase arrest. The tumor size was smaller and the number of lung metastatic nodules was less in KIF3C depletion MDA-MB-231 cell xenograft mice than in negative control group. These results suggested that high expression of KIF3C in breast cancer may be associated with the tumor progression and metastasis.
The cellular and molecular mechanisms responsible for human idiopathic ascending aortic aneurysm (IAAA) remain unknown. Matrix metalloproteinase-2 (MMP-2) is a key enzyme for the degradation of extracellular matrix in aneurysmal walls. The aim of this study was to elucidate the role of the angiotensin II (Ang II) pathway in MMP-2 induction in IAAA aortic walls. Quantitative polymerase chain reaction and western blot analysis were used to compare the MMP-2 mRNA and protein levels in ascending aortic specimens with those in IAAA patients (n = 10) and heart transplant donors (n = 5) without any aortopathy. It was found that MMP-2 expression was significantly increased, which was associated with elastic lamellae disruption in IAAA walls. Additionally, the expression levels of angiotensinogen (AGT) and Ang II in the ascending aortic tissues from individuals with and without IAAAs were detected by western blot analysis and radioimmunoassay, respectively. The results demonstrated that the expressions of AGT and Ang II protein were significantly increased in the ascending aortic tissues of IAAA patients. Furthermore, whether Ang II induces MMP-2 expression was investigated using human IAAA walls ex vivo culture. It was found that exogenous Ang II increased the MMP-2 expression in a dose-dependent manner, which was completely inhibited by the Ang II type 1 receptor (AT1R) inhibitor candesartan and was mediated by c-Jun N-terminal kinase (JNK) activation. Taken together, these results indicate that Ang II can induce an increase of MMP-2 expression via AT1R and JNK in ex vivo cultured IAAA aortic walls, and suggest that angiotensin receptor blocker (ARB) drugs and JNK inhibitors have the potential in the prevention or treatment of IAAAs.
In this study, we hypothesized that angiotensin II (Ang II) induces matrix metalloproteinase 2 (MMP-2) upregulation in aneurysmal smooth muscle cells (ASMCs) derived from ascending thoracic aortic aneurysms (ATAAs). We compared MMP-2 protein levels in ascending aortic specimens using Western blot and plasma concentrations by enzyme-linked immunosorbent assay between ATAA (n = 40) and coronary heart disease patients (n = 40). Additionally, the protein level of angiotensinogen (AGT) in the ascending aorta and the plasma concentration of Ang II were detected by Western blot and radioimmunoassay, respectively, in ATAA and coronary heart disease patients. In ATAA patients, Ang II and MMP-2 plasma levels were significantly increased (P < 0.05). Additionally, AGT and MMP-2 protein levels in the aorta of ATAA patients were higher (P < 0.01). Enhanced AGT suggested that the amount of Ang II in aneurysmal aorta specimens may be also increased, which was confirmed by immunofluorescent staining for Ang II. Moreover, we investigated the effect of Ang II on MMP-2 upregulation by ASMCs and determined the Ang II receptors and intracellular signaling pathways that are involved. Our results showed that treatment with Ang II significantly increased the expression of MMP-2 through the Ang II type 1 receptor (AT1R) and activated the 3 major mitogen-activated protein kinases (MAPKs), JNK, ERK1/2, and p38 MAPK. In conclusion, these results indicate that Ang II can induce MMP-2 expression elevation through AT1R and MAPK pathways in ASMCs and suggest that there is therapeutic potential for angiotensin receptor blocker drugs and MAPK inhibitors in the prevention and treatment of ATAAs.
Human urinary bladder cancer (UBC) is the fourth most common cancer and the eighth most common cause of cancer death in the USA. High mobility group box 3 (HMGB3), a member of a family of proteins containing one or more high mobility group DNA binding motifs, was reported to be overexpressed in a variety of human cancers. However, the expression and role of HMGB3 in human UBC remains unclear. Here, we found that UBC patients had upregulated HMGB at both mRNA and protein levels. Immunochemistry (IHC) evaluation of HMGB3 expression in 113 UBC clinical specimens showed that high expression of HMGB3 had positive correlation with UBC tumor size (P = 0.019), tumor WHO grade (P = 0.031), stage (P = 0.028), and lymph node metastasis (P = 0.017). Moreover, patients with higher HMGB3 expression showed a poorer overall survival rate than those with relatively low HMGB3 (P = 0.0079, log-rank test). Multivariate analysis revealed that HMGB3 expression is an independent prognostic marker. The UBC cancer cell proliferation and migration ability were measured by 3-(4, 5-dimethylthiazol-2-yl)-2, 5-diphenyltetrazolium bromide (MTT) and wound healing assays, respectively. RNA interference of HMGB3 in UBC cell lines inhibited cancer cell growth and migration, along with the downregulation of PCNA and MMP2 protein levels. In sum, our data suggests HMGB3 may serve as an important oncoprotein and indicate that overexpression of HMGB3 in UBC could be used as a potential prognostic marker.
FOXP3 is known as a master control of regulatory T cells with recently studies indicating its expression in several tumor cells. In order to study the precise role of FOXP3 in cholangiocarcinoma, FOXP3 was knocked down in cholangiocarcinoma cell lines. Down regulation of FOXP3 inhibits tumor cell invasion by reducing the quantity of MMP-9 and MMP-2. With FOXP3 knocking down, IL-10 and TGF-β1 secreted by cancer cells diminishes and the cell survival of T cells is significant up-regulation. These results suggest that FOXP3 plays an important role in tumor malignant phenotype, especially the invasion and immune escape.
FoxM1 is a specific transcription factor that has an important function in aggressive human carcinomas, including cervical cancer. However, the specific function and internal molecular mechanism in cervical cancer remain unclear. In this study, RNAi-mediated FoxM1 knockdown inhibited cell growth. This process also decreased the migration and invasion activities of HeLa cells in vitro. Downregulation of FoxM1 inhibited tumor growth and angiogenesis in vivo. In addition, the expressions of uPA, matrix metalloproteinase (MMP)-2, MMP-9 and VEGF were significantly decreased in vitro and in vivo. These results suggested that the inactivation of FoxM1 could be a novel therapeutic target for cervical cancer treatment.
The phosphatase and tensin homolog (PTEN) gene, an important tumor-suppressor gene, has been demonstrated to have the potential for inhibiting proliferation, migration and invasion in various types of cancer cells. The aim of the present study was to investigate the effect of PTEN expression on osteosarcoma (OS) cells. The wild-type PTEN plasmid was transfected into OS U2-OS cells. The effects of PTEN on the adhesion, migration and invasion of U2-OS cells were evaluated by cell adhesion analysis, in vitro scratch and Transwell assays, respectively. The levels of MMP-2 and MMP-9, and focal adhesion kinase (FAK) protein regulated by PTEN were detected via western blot analysis. Meanwhile, the level of intracellular FAK phosphorylation was observed. The results from the present study showed that overexpression of PTEN transcription and protein were observed in U2-OS cells following PTEN transfection. Furthermore, the migration, invasion and adhesion capabilities of the cells with PTEN transfection were significantly decreased compared to these capacities in the cells without PTEN. Meanwhile, it was shown that there was downregulation of MMP-9, FAK and p-FAK concomitant with the elevation of the intracellular PTEN level. It is therefore evident that the upregulation of PTEN may attenuate the adhesion, migration and invasion capabilities of OS cells. The mechanisms of the effects of PTEN on OS cells may be correlated with a reduction in the related genes by PTEN regulation.
Sophoricoside (SOPH) is an isoflavone glycoside isolated from the fruits of Sophora japonica. Since its first isolation in 1961, there are rare findings about the effects of SOPH on reproductive system. In the present study, the pregnant mice administrated by different doses of SOPH were used to explore the effect of SOPH on embryo implantation, especially on the endometrial receptivity. The statistical results showed that the number of implanted embryos was gradually declining along the increasing dose of SOPH. When the administrated dose of SOPH was 600 mg/kg per day, great changes were observed in the exposed uterine morphology and up-regulated progesterone receptor (PR) and down-regulated estrogen receptor α (ERα), E-cadherin, matrix metalloproteinase-2 (MMP-2) and integrin β3 were also found in SOPH-exposed uterine. These findings demonstrated that SOPH exposure reduced the number of implanted embryos in a dose-dependent manner and failed the embryo implantation through altering the morphology of uterine and compromising the endometrial receptivity.
Blood-brain barrier (BBB) disruption and brain edema formation play important roles in the secondary neuronal death and neurological dysfunction induced by intracerebral hemorrhage (ICH). Poloxamer 188 (P188), a multiblock copolymer surfactant, has been shown to be capable of sealing damaged cell membranes and decrease neuronal cell death. In this study, we explored whether P188 had a protective effect against ICH and its underlying mechanisms. Male ICR mice were subjected to infusion of type IV collagenase (to induce ICH) of saline (for shams) into the left striatum. The results showed that P188-12 mg post-treatment by tail intravenous injection significantly ameliorated the neurological symptoms and brain edema, attenuated BBB permeability, and decreased cell insults and injury volume at 24 and 72 h after ICH. Furthermore, P188 maintained the protein levels of tight junction (TJ) proteins including claudin-5, occludin, and zonula occludens-1, and reversed the increases of nuclear factor-kappaB (NF-κB), matrix metalloproteinase (MMP)-2, and MMP-9 protein expression at 72 h post ICH. Immunofluorescence showed P188 treatment rearranged the structure of TJ proteins in a continuous and linear pattern. Therefore, the present study concludes that P188 can protect against ICH, and the protective effect was associated with preventing BBB disruption through NF-κB-MMPs-mediated TJ proteins degradation.
Accumulating evidences implicate that ribonuclease inhibitor (RI) plays a suppressing role in cancer development. However, the mechanisms underlying antitumor of RI remain largely unknown. Epithelial-mesenchymal transition (EMT) is regarded as a key event in tumor progression. The reports have demonstrated that EMT was implicated in metastasis of bladder cancer. Therefore, we suppose that RI might involve regulating EMT of bladder cancer. Here bladder cancer T24 cells were transfected with pGensil-1-siRNA-RI vectors. HE staining, living cell observation, Phalloidine-FITC staining of microfilament, cell adhesion, scratch migration, and Matrigel invasion were examined respectively. RI expression and colocalization with ILK were detected using confocal microscope. Proteins associated with EMT were determined with Western blotting and immunohistochemistry in vivo and in vitro. Effects of RI expression on tumor growth, metastasis and EMT related proteins in BALB/C nude mouse and clinical human bladder cancer specimens were valued with histological, immunohistochemical and immunofluorescent examination respectively. We demonstrated that down-regulating RI increased cell proliferation, migration and invasion, changed cell morphology and adhesion, and rearranged cytoskeleton by inducing EMT and ILK signaling pathway in bladder cancer cells. In addition, we showed that down-regulating RI promoted tumorigenesis and metastasis of bladder cancer in vivo. Finally, we found that bladder cancer with invasive capability had higher Vimentin, Snail, Slug and Twist as well as lower E-cadherin and RI expression in clinical human specimens. Our results suggest that RI could play a novel role in inhibiting metastasis of bladder through regulating EMT and ILK signaling pathway.
Tumor invasion and migration obstructs the treatment and prognosis of cancer. In this research, we investigated the effect of oroxylin A, a natural compound extracted from Scutellaria radix, the root of Scutellaria baicalensis, on inhibition of the invasion and migration of three different tumor cell lines: MCF-7, DU145, and HepG2. The results suggested that oroxylin A could inhibit hypoxia-induced migration and invasion of the three cell lines mentioned above. To study the detailed mechanisms, studies were carried out on MCF-7 cells and it was found that oroxylin A could regulate the expression of related markers in MCF-7 cells including E-cadherin, N-cadherin, and Vimentin. It was also found that oroxylin A inhibited the hypoxia-induced invasion and migration of MCF-7 cells by suppressing the Notch pathway. Oroxylin A inhibited N1ICD translocating to the nucleus and binding to epithelial-mesenchymal transition-related transcription factor Snail, thus suppressing the invasion and migration of MCF-7 cells. Therefore, oroxylin A is expected to be a promising candidate for antimetastasis treatment through suppression of the hypoxia-induced Notch pathway.
A liposome system modified with chlorotoxin (ClTx), a scorpion venom peptide previously utilized for targeting brain tumors, was established. Its targeting efficiency and antimetastasis behavior against metastatic breast cancer highly expressed MMP-2, the receptor of ClTx, were investigated. 4T1, a metastatic breast cancer cell line derived from a murine breast tumor, was selected as the cell model. As results, the ClTx-modified liposomes displayed specific binding to 4T1 as determined by flow cytometry and confocal imaging. The cytotoxicity assay revealed that the ClTx modification increased the toxicity compared with nonmodified liposomes. In addition, the modified liposomes also exhibited high in vivo targeting efficiency in the BALB/c mice bearing 4T1 tumors. Importantly, this system inhibited the growth of metastatic tumor and prevented the incidence of lung metastasis in mice bearing 4T1 tumors with only low systemic toxicity. The data obtained from the in vitro and in vivo studies confirmed that the ClTx-modified liposomes increased the drug delivery to metastatic breast cancers. This study proved that the ClTx-modified liposomes had targeting ability to metastatic breast cancer in addition to brain cancer, and displayed an obvious antimetastasis effect. Generally, it may provide a promising strategy for metastatic breast cancer therapy.
Isocitrate dehydrogenase (IDH) is of great importance in cell metabolism and energy conversion. IDH mutation in glioma cells is reported to be associated with an increased overall survival. However, effects biological behavior of therapy of gliomas are unclear. Here, we investigated the influence of wild-type and mutated IDH genes on glioma cell biological behavior and response to chemotherapy. Relevant mechanisms were further explored. We designed our study on the background of the IDHR132H mutation. Stable cell lines were constructed by transfection. The CCK-8 method was used to assess cell proliferation, flow cytometry for the cell cycle and cell apoptosis, and the transwell method for cell invasion. Nude mouse models were employed to determine tumorigenesis and sensitivity to chemotherapy. Western blotting was used to detect relevant protein expression levels. We found that overexpression of wild IDH1 gene did not cause changes in the cell cycle, apoptosis and invasion ability. However, it resulted in chemotherapy resistance to a high dose of temozolomide (TMZ) in vivo and in vitro. The IDH1 mutation caused cell cycle arrest in G1 stage and a reduction of proliferation and invasion ability, while raising sensitivity to chemotherapy. This may provide an explanation for the better prognosis of IDH1 mutated glioma patients and the relative worse prognosis of their wild-type IDH1 counterparts. We also expect IDH1 mutations may be optimized as new targets to improve the prognosis of glioma patients.
Matrix metalloproteinase (MMP)-2 and MMP-9 have been known to play the role of essential mediators in angiogenesis. Non-invasive in vivo imaging approach using imaging probes is a potential method of detecting MMP activity in living animals, wherein imaging probes must include the characteristics of non-toxicity, specific targetability, and reasonable signal intensity. Here, we developed MMP-specific and self-quenched human serum albumin (HSA)-based (MMP-HSA) nanoprobes for non-invasive optical imaging of MMP activity during angiogenesis in the mouse hindlimb ischemia model. MMP-specific fluorogenic peptide probes, which were self-quenched with a near-infrared fluorophore and a quencher, were covalently conjugated to HSA (MMP-HSA nanoprobes). MMP-HSA nanoprobes formed stable nanoparticle structures of approximately 36 nm in diameter. Strongly self-quenched MMP-HSA nanoprobes boosted intense fluorescence signals in the presence of MMP-2 and MMP-9. Furthermore, MMP-HSA nanoprobes showed no cytotoxicity in cell culture. Importantly, intravenous injection of MMP-HSA nanoprobes provided longer blood half-life and successful non-invasive optical imaging of MMP activity during angiogenesis in the mouse hindlimb ischemia model. In addition, the MMP activity visualized by MMP-HSA nanoprobes was consistent with the results of zymography, Western blot, and immunohistochemistry. MMP-HSA nanoprobes may be useful for monitoring of the initial process of angiogenesis through non-invasive MMP imaging.
Integrin-linked kinase (ILK) is a multifunctional serine/threonine kinase. Accumulating evidences suggest that ILK are involved in cell-matrix interactions, cell proliferation, invasion, migration, angiogenesis and Epithelial-mesenchymal transition (EMT). However, the underlying mechanisms remain largely unknown. EMT has been postulated as a prerequisite for metastasis. The reports have demonstrated that EMT was implicated in metastasis of oral squamous cell carcinomas. Therefore, here we further postulate that ILK might participate in EMT of tongue cancer. We showed that ILK siRNA inhibited EMT with low N-cadherin, Vimentin, Snail, Slug and Twist as well as high E-cadherin expression in vivo and in vitro. We found that knockdown of ILK inhibited cell proliferation, migration and invasion as well as changed cell morphology. We also demonstrated that ILK siRNA inhibited phosphorylation of downstream signaling targets Akt and GSK3β as well as reduced expression of MMP2 and MMP9. Furthermore, we found that the tongue tumor with high metastasis capability showed higher ILK, Vimentin, Snail, Slug and Twist as well as lower E-cadherin expression in clinical specimens. Finally, ILK siRNA led to the suppression for tumorigenesis and metastasis in vivo. Our findings suggest that ILK could be a novel diagnostic and therapeutic target for tongue cancer.
Human ribonuclease inhibitor (RI) is a cytoplasmic acidic protein possibly involved in biological functions other than the inhibition of RNase A and angiogenin activities. We have previously shown that RI can inhibit growth and metastasis in some cancer cells. Epithelial-mesenchymal transition (EMT) is regarded as the beginning of invasion and metastasis and has been implicated in the metastasis of bladder cancer. We therefore postulate that RI regulates EMT of bladder cancer cells. We find that the over-expression of RI induces the up-regulation of E-cadherin, accompanied with the decreased expression of proteins associated with EMT, such as N-cadherin, Snail, Slug, vimentin and Twist and of matrix metalloprotein-2 (MMP-2), MMP-9 and Cyclin-D1, both in vitro and in vivo. The up-regulation of RI inhibits cell proliferation, migration and invasion, alters cell morphology and adhesion and leads to the rearrangement of the cytoskeleton in vitro. We also demonstrate that the up-regulation of RI can decrease the expression of integrin-linked kinase (ILK), a central component of signaling cascades controlling an array of biological processes. The over-expression of RI reduces the phosphorylation of the ILK downstream signaling targets p-Akt and p-GSK3β in T24 cells. We further find that bladder cancer with a high-metastasis capability shows higher vimentin, Snail, Slug and Twist and lower E-cadherin and RI expression in human clinical specimens. Finally, we provide evidence that the up-regulation of RI inhibits tumorigenesis and metastasis of bladder cancer in vivo. Thus, RI might play a novel role in the development of bladder cancer through regulating EMT and the ILK signaling pathway.
Di-(2-ethylhexyl)-phthalate (DEHP) is a ubiquitous environmental pollutant and endocrine disruptor (ED) that causes serious adverse effects on animal and human health. The harmful effects of DEHP on human reproduction are increasingly recognized, especially in women. However, it is not known how endometrial receptivity and embryo implantation, which play important roles in the establishment of pregnancy, are affected by DEHP. This study was aimed towards investigating the effects of DEHP on endometrial receptivity and embryo implantation in pregnant mice. The pregnant mice received DEHP at 0, 250, 500 and 1000 mg/kg/day from day 1 (D1) of gestation until sacrifice. Administration of DEHP led to compromised endometrial receptivity and decreased number of implantation sites. The mRNA and protein expression levels of ERα, PR and E-cadherin, but not those of HoxA10 and MMP-2, were up-regulated by DEHP in the mouse endometrium. The results further suggested that DEHP disrupts the MAPK and NF-κB signaling pathways. This was maybe one of paths which influenced the E-cadherin expression. In conclusion, DEHP reduced endometrial receptivity and impaired embryo implantation by influencing the expression of hormone receptors and E-cadherin. Therefore, determining the full extent of the hazards of DEHP to human reproduction will be vital to developing and implementing effective protective measures.
The aim of this study was to determine the disease activity index (DAI) and the colonic mucosa damage index (CMDI), and to detect the colonic mucosal expression levels of matrix metalloproteinase-2 (MMP-2) and tumor necrosis factor-α (TNF-α) in rats with ulcerative colitis (UC). We also aimed to investigate the protective role of Etiasa in UC. Sprague Dawley (SD) rats were randomly divided into three groups: the control, an Etiasa-treated group and a UC model group. Rats were sacrificed on days 14, 21, 35 or 56 following the administration of treatment by enema and the DAI, CMDI and colonic expression levels of MMP-2 and TNF-α were assessed. In the UC model group, the DAI and CDMI scores and the colonic expression levels of MMP-2 and TNF-α increased significantly compared with the control at all timepoints, and were also significantly higher than those in the Etiasa-treated group. In conclusion, the expression levels of MMP-2 and TNF-α increased significantly in rats with UC. Etiasa reduces colonic mucosal damage by downregulating the expression of MMP-2 and TNF-α.
Nineteen natural compounds with diverse structures are identified as potential MMPIs using structure-based virtual screening from 4000 natural products. Hydroxycinnamic acid or analogs of natural products are important for potent inhibitory and selectivity against MMPs, and the solvent effect in the S1' pocket can affect the hydrophobic interactions and hydrogen bonds between MMPIs and MMPS, making MMPIs exhibit certain selectivity for a specific MMP isoenzyme. Furthermore, compound 5 can reduce the expression of both MMP-2 and active-MMP-9, and suppress the migration of MDA-MB-231 tumor cell in a wound healing assay, which may be further developed as an anticancer agent.
Human ribonuclease inhibitor (RI) is a cytoplasmic acidic protein. RI is constructed almost entirely of leucine rich repeats, which might be involved in unknown biological effects except inhibiting RNase A and angiogenin activities. We previously reported that up-regulating RI inhibited the growth and metastasis of melanoma cells. Epithelial-mesenchymal transition (EMT) is a critical event of cancer cells that triggers invasion and metastasis. However, the role of RI in the EMT process remains unknown. Here we hypothesize that RI might inhibit melanoma invasion and metastasis by regulating EMT. We found that over-expression of RI induced up-regulation of E-cadherin, accompanied with decreased expressions of proteins associated with EMT such as N-cadherin, Snail, Slug, Vimentin and Twist both in vitro and in vivo. Furthermore, RI restrained matrix metalloproteinase MMP-2 and MMP-9 secretions in B16 and B16-F10 melanoma cells. In addition, we also found that up-regulation of RI inhibited cell proliferation, migration and invasion as well as changed cell morphology, adhesion and rearranged cytoskeleton in vitro. Finally, the effects of RI on phenotype and invasiveness translated into suppressing metastasis by the experimental metastasis models of melanoma with lighter lung weight, a fewer metastasis nodules and a lower incidence rate, with respect to the control groups. Taken together, our data highlight, for the first time, that RI plays a novel role in inhibiting development and progression of murine melanoma cells through regulating EMT. These results suggest that RI could be a therapeutic target protein for melanoma and may be of biological importance.
P21-activated kinase 4 (PAK4), a serine/threonine protein kinase, has involved in the regulation of cytoskeletal reorganization, cell proliferation, gene transcription, oncogenic transformation and cell invasion. Moreover, PAK4 overexpression, genetic amplification and mutations were detected in a variety of human tumors, which make it potential therapeutic target. In this paper we found that LCH-7749944, a novel and potent PAK4 inhibitor, effectively suppressed the proliferation of human gastric cancer cells through downregulation of PAK4/c-Src/EGFR/cyclin D1 pathway. In addition, LCH-7749944 significantly inhibited the migration and invasion of human gastric cancer cells in conjunction with concomitant blockage of PAK4/LIMK1/cofilin and PAK4/MEK-1/ERK1/2/MMP2 pathways. Interestingly, LCH-7749944 also inhibited the formation of filopodia and induced cell elongation in SGC7901 cells. Importantly, LCH-7749944 caused successful inhibition of EGFR activity due to its inhibitory effect on PAK4. Taken together, these results provided novel insights into the development of PAK4 inhibitor and potential therapeutic strategies for gastric cancer.
Arsenic trioxide (As(2)O(3)) has been shown to inhibit invasion/migration in cancer cells. However, the underlying mechanism is poorly understood. To identify the role of As(2)O(3) in regulating invasion/migration activity in human gastric cancer SGC-7901 cells, the effects of As(2)O(3) on cell invasion/migration activity, the expression of cyclooxygenase-2 (Cox-2), prostaglandin E2 (PGE2), thromboxane B2 (TXB2), leukotriene B4 (LTB4), and matrix metalloproteinase-2 (MMP-2) and intracellular reactive oxygen species (ROS) were examined. Furthermore, N-acetyl-l-cysteine (NAC, a radical scavenger) and celecoxib (a Cox-2 inhibitor) were used to explore the molecular mechanism. The results demonstrated that As(2)O(3) (1 and 2 μmol/L) inhibited invasion/migration activity in SGC-7901 cells at 24 h and suppressed the expression of Cox-2, PGE2 and MMP-2 (P < 0.05), whereas the same treatment had no significant effect on TXB2 and LTB4 expression. In contrast, intracellular ROS were increased (P < 0.05). Moreover, NAC eliminated the excessive ROS and restored the expression of Cox-2 and MMP-2 and invasion/migration activity in As(2)O(3)-treated cells (P < 0.05). These results suggest that ROS may be a critical factor in regulating the invasion/migration process. Moreover, celecoxib significantly decreased Cox-2, MMP-2 and PGE2 expression and inhibited invasion/migration activity in As(2)O(3)-treated cells (P < 0.05), indicating that As(2)O(3) inhibits invasion/migration by regulating the expression of Cox-2/PGE2/MMP-2. In conclusion, these results suggest that increased ROS play a critical role in inhibiting invasion/migration by suppressing the Cox-2/MMP-2 pathway in As(2)O(3)-treated SGC-7901 cells and regulating intracellular ROS levels may be a promising strategy in gastric cancer therapy.
Resveratrol is an antioxidant and anti-inflammatory polyphenol. Periodontitis is induced by oral pathogens, where a systemic inflammatory response accompanied by oxidative stress is the major event initiating disease. We investigated how resveratrol modulates cellular responses and the mechanisms related to this modulation in lipopolysaccharide (LPS)-stimulated human gingival fibroblasts (hGFs). We also explored whether resveratrol protects rats against alveolar bone loss in an experimental periodontitis model. Periodontitis was induced around the first upper molar of the rats by applying ligature infused with LPS. Stimulating hGFs with 5μg/ml LPS augmented the expression of cyclooxygenase-2, matrix metalloproteinase (MMP)-2, MMP-9, and Toll-like receptor-4. LPS treatment also stimulated the production of reactive oxygen species (ROS) and the phosphorylation of several protein kinases in the cells. However, the expression of heme oxygenase-1 (HO-1) and nuclear factor-E2 related factor 2 (Nrf2) was inhibited by the addition of LPS. Resveratrol treatment almost completely inhibited all of these changes in LPS-stimulated cells. Specifically, resveratrol alone augmented HO-1 induction via Nrf2-mediated signaling. Histological and micro-CT analyses revealed that administration of resveratrol (5mg/kg body weight) improved ligature/LPS-mediated alveolar bone loss in rats. Resveratrol also attenuated the production of inflammation-related proteins, the formation of osteoclasts, and the production of circulating ROS in periodontitis rats. Furthermore, resveratrol suppressed LPS-mediated decreases in HO-1 and Nrf2 levels in the inflamed periodontal tissues. Collectively, our findings suggest that resveratrol protects rats from periodontitic tissue damage by inhibiting inflammatory responses and by stimulating antioxidant defense systems.
STATEMENT OF SIGNIFICANCE:
The aims of this study were to investigate how resveratrol modulates cellular responses and the mechanisms related to this modulation in lipopolysaccharide (LPS)-stimulated human gingival fibroblasts (hGFs) and protects rats against alveolar bone disruption in an experimental periodontitis model. Our findings suggest that resveratrol protects rats from periodontitic tissue damage by inhibiting inflammatory responses and by stimulating antioxidant defense systems. On the basis of our experiment studies, we proposed that resveratrol could be used as novel bioactive materials or therapeutic drug for the treatment of periodontitis or other inflammatory bone diseases like osteoporosis, arthritis etc. Furthermore, it could be also used for the modification or coating of implant materials as an antiinflammatory molecules which will help to accelerate bone formation. There are a few of reports suggesting antioxidant and anti-inflammatory potentials of resveratrol. However, our results highlight the cellular mechanisms by which resveratrol inhibits LPS-mediated cellular damages using human-originated gingival fibroblasts and also support the potential of resveratrol to suppress periodontitis-mediated tissue damages. We believe that the present findings might improve a clinical approach of using of resveratrol on human, although further detailed experiments will be needed.
Copyright © 2015 Acta Materialia Inc. Published by Elsevier Ltd. All rights reserved.
BACKGROUND/AIMS:
Annexin II receptor (AXIIR) is able to mediate Annexin II signal and induce apoptosis, but its role in angiogenesis remains unclear. This study tries to investigate the role of AXIIR in angiogenesis and the plausible molecular mechanism.
METHODS/RESULTS:
RNA interference technology was used to silence AXIIR, and the subsequent effects in vitro and in vivo were evaluated thereafter. Our data indicated that human umbilical vein endothelial cells (HUVECs) expressed AXIIR and knockdown of AXIIR significantly inhibited HUVECs proliferation, adhesion, migration, and tube formation in vitro and suppressed angiogenesis in vivo. Furthermore, AXIIR siRNA induced cell arrest in the S/G2 phase while had no effect on cell apoptosis. We found that these subsequent effects might be via suppressing the expression of matrix metalloproteinase 2and matrix metalloproteinase 9.
CONCLUSION:
AXIIR participates in angiogenesis, and may be a potential therapeutic target for angiogenesis related diseases.
© 2015 S. Karger AG, Basel.
AIM:
To examine epigenetic changes and the function of HOXA11 in human gastric cancer (GC).
MATERIALS & METHODS:
Seven GC cell lines, five cases of normal gastric mucosa and 112 cases primary GC samples were used in this study.
RESULTS:
Expression of HOXA11 and lack of promoter region methylation were found in NCI-N87, MKN45, BGC823 and HGC27 cells. Loss of expression and complete methylation were found in AGS gastric cancer cells. Reduced expression and partial methylation were found in MGC803 and SGC7901 cells. Restoration of HOXA11 expression was induced by 5-aza-2'-deoxycytidine. HOXA11 was methylated in 81.25% (91/112) of primary GCs. The presence of methylation was associated with male gender, tumor size, tumor differentiation and lymph node metastasis (all p < 0.05). Restoration of HOXA11 expression reduced cell proliferation, invasion, migration and induced apoptosis and G2/M phase arrest. HOXA11 was found to inhibit Wnt signaling by upregulating NKD1 expression.
CONCLUSION:
Epigenetic silencing of HOXA11 promotes GC proliferation, migration and invasion through activation of Wnt signaling.
BACKGROUND:
Fangchinoline as a novel anti-tumor agent has been paid attention in several types of cancers cells except lung cancer. Here we have investigated the effect of fangchinoline on A549 cells and its underlying mechanism.
PURPOSE:
The purpose of this work was to study the effect of fangchinoline on A549 cells.
METHODS:
Four lung cancer cell lines (A549, NCI-H292, NCI-H446, and NCI-H460) were exposed to varying concentrations (10-40 μmol/l) of fangchinoline to observe the effect of fangchinoline on the four lung cancer cell lines and to observe the changes of the lung cancer cell on proliferation, apoptosis, and invasion.
RESULTS:
Fangchinoline effectively suppressed proliferation and invasion of A549 cell line but not NCI-H292, NCI-H446, and NCI-H460 cell lines by inhibiting the phosphorylation of FAK (Tyr397) and its downstream pathways, due to the significant differences of Fak expression between A549 and the other three cell lines. And all FAK-paxillin/MMP2/MMP9 pathway, FAK-Akt pathway, and FAK-MEK-ERK1/2 pathway could be inhibited by fangchinoline.
DISCUSSION:
Fangchinoline effectively suppressed proliferation and invasion of A549 cell line by inhibiting the phosphorylation of FAK (Tyr397) and its downstream pathways.
CONCLUSION:
Fangchinoline could inhibit the phosphorylation of FAK(p-Tyr397), at least partially. Fangchinoline as a kinase inhibitor targets FAK and suppresses FAK-mediated signaling pathway and inhibits the growth and the invasion in tumor cells which highly expressed FAK such as A549 cell line.
PURPOSE:
Oxymatrine (matrine oxide, matrine N-oxide, matrine 1-oxide) is one of the quinolizidine alkaloid compounds extracted from the root of Sophora flavescens (a traditional Chinese herb). Oxymatrine has been known for its chemoresistance and cytotoxic effects on various cancer cells, but the mechanism underlying has not been explored. We study the mechanism of oxymatrine on gastric cells.
METHODS:
We observed the changes of proliferation, apoptosis and invasion in human gastric cells by detecting the signaling pathway in which oxymatrine plays role.
RESULTS:
These results showed that oxymatrine inhibited the proliferation and invasion of gastric cells through inhibition of EGFR/Cyclin D1/CDK4/6, EGFR/Akt and MEK-1/ERK1/2/MMP2 pathway by inhibiting EGFR(p-Tyr845). In addition to inducing gastric cells apoptosis, oxymatrine significantly inhibited the migration and invasion of human gastric cancer cells by decreasing phospho-Cofilin (Ser3) and phospho-LIMK1 (Thr508) without changing the total Cofilin and LIMK1 expression.
CONCLUSION:
Oxymatrine effectively suppressed the phosphorylation of EGFR (Tyr845), and EGFR was the target of oxymatrine.
AIMS:
It has been reported that glucagon-like peptide-1 (GLP-1) agents are associated with an increased risk of pancreatic cancer in patients with type 2 diabetes. Reports have indicated that GLP-1 promotes pancreatic metaplasia and premalignant lesions. The aims of this study were to determine the effects of GLP-1-based therapy on pancreatic cancer cells.
METHODS:
Immunohistochemistry was used to investigate GLP-1 receptor (GLP-1R) expression in 30 human pancreatic cancer tissues. We also analysed associated clinicopathological data and each patient's prognosis. Two human pancreatic cancer cell lines were used to evaluate the in vitro effects of the GLP-1R agonist liraglutide on cell growth, migration and invasion. Mouse xenograft models of human pancreatic cancer were established to evaluate the effects of liraglutide in vivo.
RESULTS:
Human pancreatic cancer tissues showed lower levels or a lack of GLP-1R expression when compared with levels in the tumour-adjacent pancreatic tissues. Negative GLP-1R expression occurred more frequently in advanced tumours with larger diameters and lymphatic metastasis, and was associated with a poor prognosis. GLP-1R activation with liraglutide inhibited tumourigenicity and metastasis of human pancreatic cancer cells in vitro and in vivo. Akt activation was dose-dependently inhibited by liraglutide, and the PI3K inhibitors, LY294002 and wortmannin, displayed similar suppressive effects to liraglutide in human pancreatic cancer cells.
CONCLUSIONS:
GLP-1R activation has an antitumour effect on human pancreatic cancers via inhibition of the PI3K/Akt pathway. This finding suggests that GLP-1-based therapies may be beneficial, rather than harmful, in treating type 2 diabetic patients with pancreatic cancer.
© 2014 John Wiley & Sons Ltd.
AIMS:
Given the importance of inflammation in the onset and progression of diabetic cardiomyopathy, we investigated the potential protective effects of triptolide, an anti-inflammatory agent, in streptozotocin-induced diabetic rat model and in H9c2 rat cardiac cells exposed to high glucose.
METHODS AND RESULTS:
Diabetic rats were treated with triptolide (100, 200, or 400 μg/kg/day respectively) for 6 weeks. At the end of this study, after cardiac function measurements were performed, rats were sacrificed and their hearts were harvested for further histologic and molecular biologic analysis. Enhanced activity and expression of nuclear factor-kappaB (NF-κB) p65 in diabetic hearts were associated with increased inflammatory response, as demonstrated by increased pro-inflammatory cytokines, cell adhesion molecules and invading inflammatory cells, as well as increased fibrosis, in line with impaired left ventricular function. Triptolide attenuated these morpho-functional alterations. Furthermore, triptolide (20 ng/ml) also attenuated high glucose-induced inflammation in H9c2 rat cardiac cells.
CONCLUSION:
Our data demonstrate that anti-inflammatory effects of triptolide involving the NF-κB signaling pathway can improve left ventricular function under diabetic conditions, suggesting triptolide treatment might be beneficial in diabetic cardiomyopathy.
INTRODUCTION:
The cytotoxicity of resin-based sealer is influential on the inflammatory reaction and cell survival for oral periapical cells. In this study, pachymic acid as an antioxidant was investigated for the improvement of bone disturbance against AH Plus (Dentsply DeTrey GmbH, Konstanz, Germany)-induced inflammation in MC-3T3 E1 cells.
METHODS:
AH Plus was prepared according to the manufacturer's instructions. Using mouse osteoblast cells (MC-3T3 E1), a specimen of AH Plus was eluted with the culture medium for 1 day and was diluted by 30%. The cellular cytotoxicity and reactive oxygen species formation was determined by 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide and 2',7'-dichlorodihydrofluorescein diacetate with fluorescence-activated cell sorting. The secretion of proinflammatory cytokines was determined by an enzyme-linked immunosorbent assay, and the expression of inflammatory and osteogenic molecules was determined by immunoblotting.
RESULTS:
Cells with AH Plus elutes showed a decrease of cell viability and ALP activity. However, pachymic acid and N-acetyl-L-cysteine (control antioxidant) restored cell viability and ALP activity damaged by AH plus. The secretion of nitric oxide, tumor necrosis factor α, and interleukin-1β were increased in AH Plus-stimulated MC-3T3 E1 cells, but pachymic acid suppressed its production. Furthermore, pachymic acid reduced the receptor activator of nuclear factor-κB ligand, cyclooxygenase-2, matrix metalloproteinase-2 and -9, increased bone morphogenetic protein-2 and -7, and runt-related transcription factor 2 despite AH Plus stimuli. In addition, pachymic acid affected the removal effect of reactive oxygen species formation as did N-acetyl-L-cysteine. More importantly, pachymic acid inhibited nuclear factor-κB translocation.
CONCLUSIONS:
The property of pachymic acid can mitigate the unfavorable conditions induced by AH Plus stimuli. Therefore, the use of pachymic acid is suggested to prevent the complications of oral diseases such as inflammation and alveolar destruction of the oral cavity.
Copyright © 2013 American Association of Endodontists. Published by Elsevier Inc. All rights reserved.
BACKGROUND:
Castration-resistant prostate cancer (CRPC) is a leading cause of cancer-related deaths in elder men. This disease has limited therapeutic options and poor prognosis as the underlying molecular mechanisms are not clearly understood. Given the emerging roles of microRNA (miRNA) as a key regulator, we postulated that miRNA may play a significant role in CRPC formation.
METHODS:
miR-146a levels in 15 androgen-dependent prostate cancer (ADPC) tissues and 5 CRPC tissues were measured by qRT-PCR. Effects of miR-146a in cell proliferation and migration in vitro and in vivo were evaluated by MTT assay, colony formation assay, transwell migratory assay, and tumor formation assay, respectively.
RESULTS:
we found that miR-146a expression was significantly decreased in CRPC tissues compared to ADPC tissues. Functional analyses showed that ectopic overexpression of miR-146a in androgen-independent cell lines not only inhibited cell growth, colony formation, and migration in vitro, but also reduced tumorigenicity and angiogenesis in vivo. Mechanistic studies revealed that miR-146a repressed the expression of EGFR through binding to its 3'-untranslated region. Also, miR-146a inhibited the expression of MMP2, one of the most important genes in tumor progression. Moreover, downregulation of p-ERK expression significantly abrogated miR-146a-induced prostate cancer cell proliferation.
CONCLUSIONS:
Our findings suggest that ubiquitous loss of miR-146a is a critical mechanism for overexpression of EGFR in CRPC, which is crucial to better understanding the pathogenesis of CRPC.
Copyright © 2011 Wiley Periodicals, Inc.
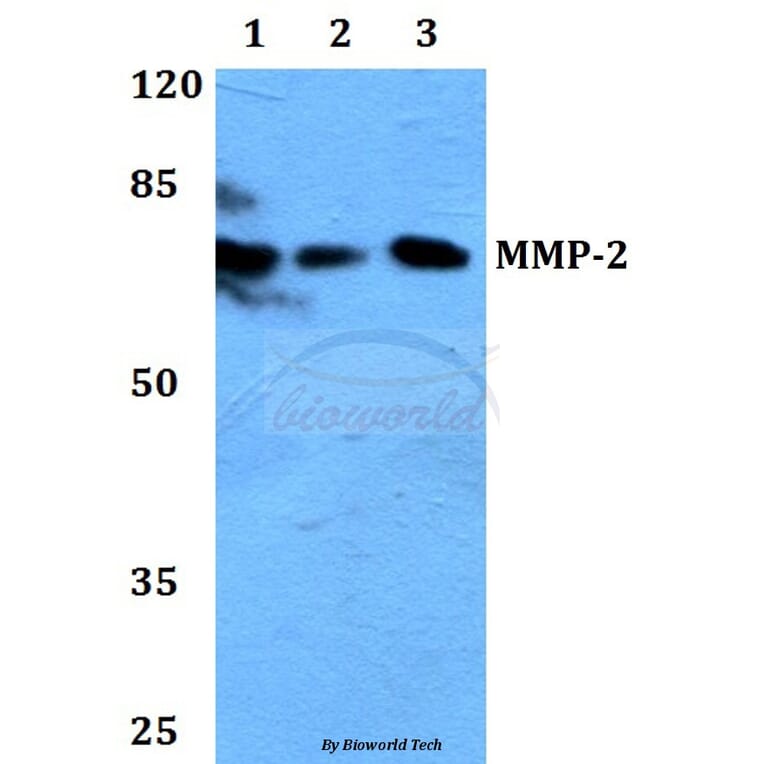







![Western Blot - Anti-MMP2 Antibody [ARC0432] (A308421) - Antibodies.com](https://cdn.antibodies.com/image/catalog/308/A308421_1.jpg?profile=product_alternative)
![Western Blot - Anti-MMP2 Antibody [MMP2/1501] (A249366) - Antibodies.com](https://cdn.antibodies.com/image/catalog/249/A249366_1.jpg?profile=product_alternative)
![Western Blot - Anti-MMP2 Antibody [MMP2/1501] - BSA and Azide free (A252546) - Antibodies.com](https://cdn.antibodies.com/image/catalog/252/A252546_1.jpg?profile=product_alternative)
