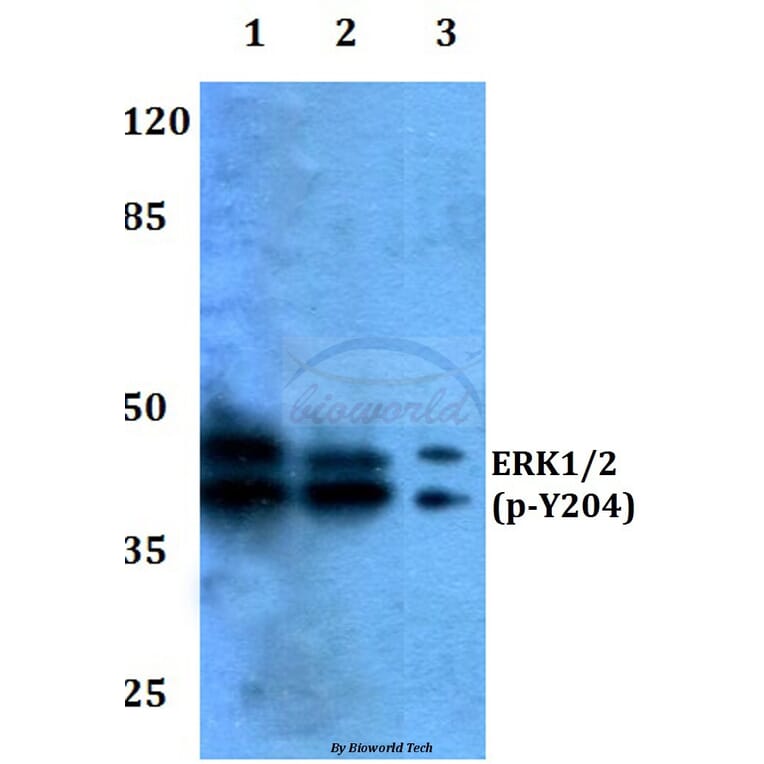
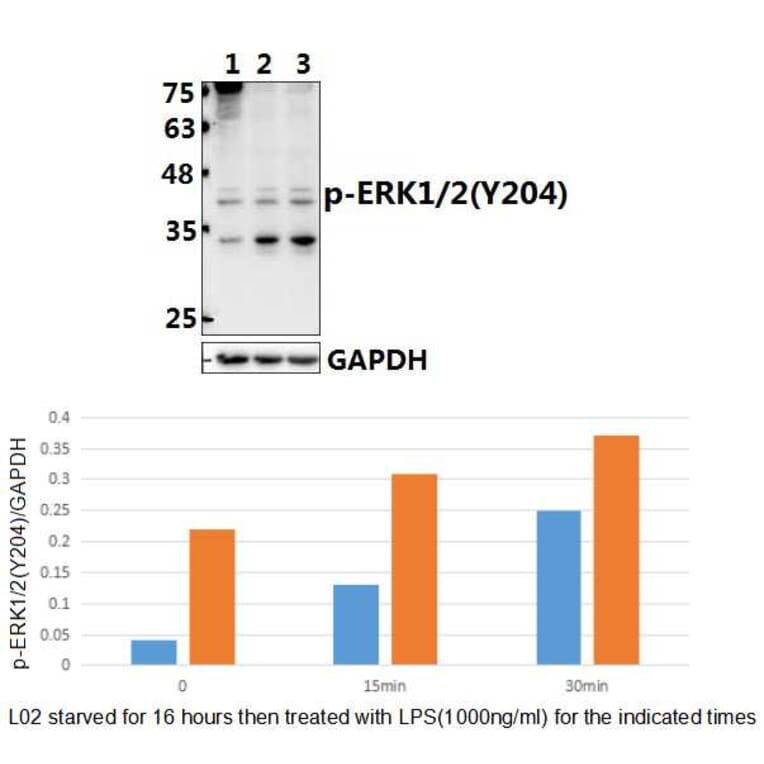
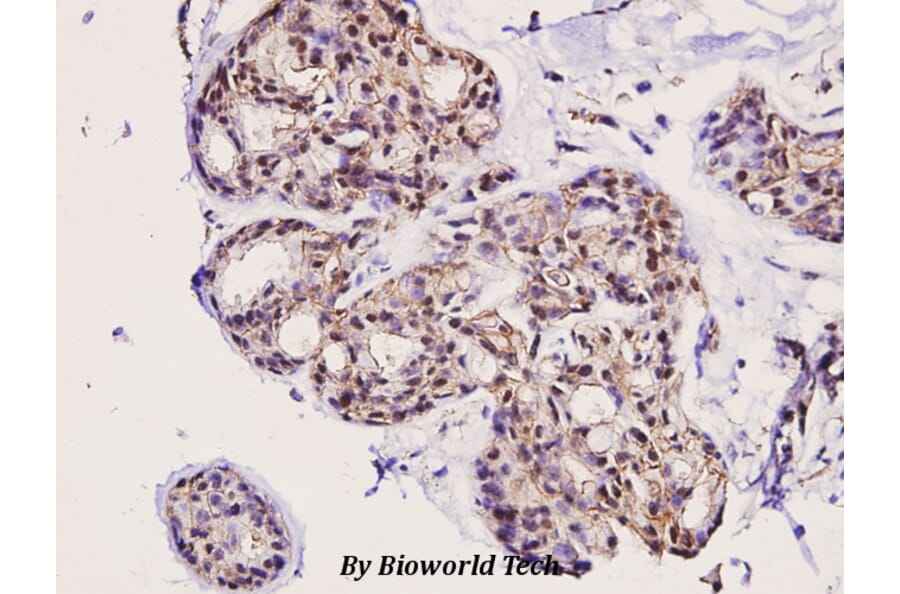
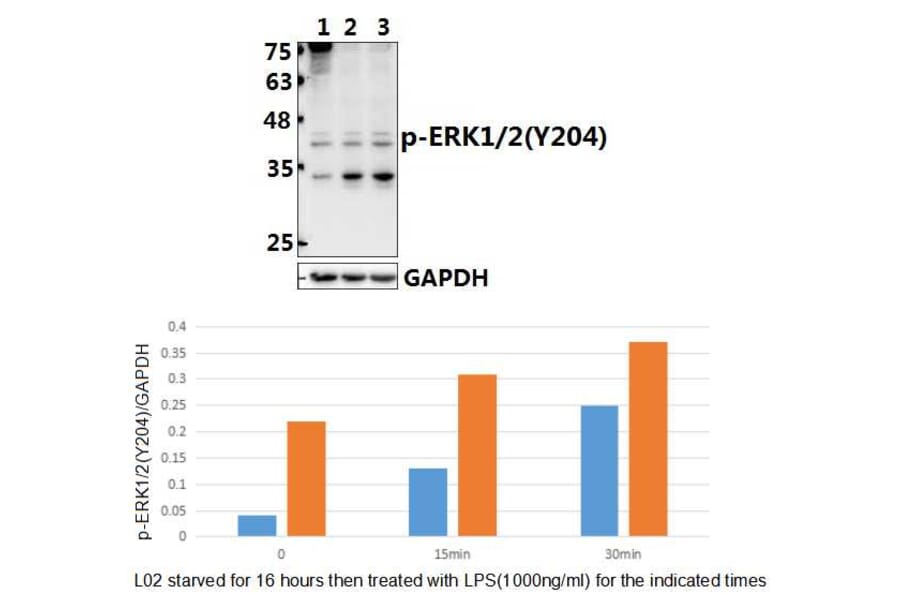
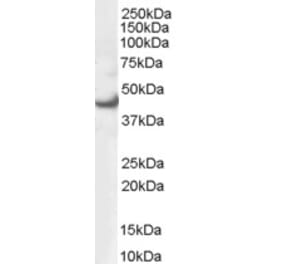
Unconjugated
IL-6, a pleiotropic cytokine, has been investigated for its role in regulating autophagy. Yet, its mechanism of action remains unclear. Here, we show that IL-6 exerted anti-autophagic effects on U937 cells through the STAT3 signaling pathway in vitro. The addition of IL-6 to starved U937 cells significantly activated the phosphorylation level of STAT3 (p-STAT3) at Tyr705 and reduced the protein levels of microtubule-associated protein 1 light chain 3 of type II (LC3-II) and Beclin 1. By immunoblotting, we also observed a positive correlation between the p-STAT3 level and Bcl-2 level. Furthermore, treatment with a STAT3 inhibitor, LLL12, or overexpression of a mutant form, STAT3Y705F, reversed the inhibitory effect of IL-6 on autophagy. Knockdown of Beclin 1 or Atg14 by siRNA and over-expression of Beclin 1 indicated the involvement of class III PI3K complex in IL-6-mediated inhibition of autophagy. Taken together, these data indicate that IL-6 inhibits starvation-induced autophagy and that p-STAT3 mediates the signal transduction from IL-6 to downstream proteins including Bcl-2 and Beclin1.
The protein tyrosine phosphatase 1B (PTP1B), a non-transmembrane protein tyrosine phosphatase, has been implicated in gastric pathogenesis. Several lines of recent evidences have shown that PTP1B is highly amplified in breast and prostate cancers. The aim of this study was to investigate PTP1B amplification in gastric cancer and its association with poor prognosis of gastric cancer patients, and further determine the role of PTP1B in gastric tumorigenesis. Our data demonstrated that PTP1B was significantly up-regulated in gastric cancer tissues as compared with matched normal gastric tissues by using quantitative RT-PCR (qRT-PCR) assay. In addition, copy number analysis showed that PTP1B was amplified in 68/131 (51.9%) gastric cancer cases, whereas no amplification was found in the control subjects. Notably, PTP1B amplification was positively associated with its protein expression, and was significantly related to poor survival of gastric cancer patients. Knocking down PTP1B expression in gastric cancer cells significantly inhibited cell proliferation, colony formation, migration and invasion, and induced cell cycle arrested and apoptosis. Mechanically, PTP1B promotes gastric cancer cell proliferation, survival and invasiveness through modulating Src-related signaling pathways, such as Src/Ras/MAPK and Src/phosphatidylinositol-3-kinase (PI3K)/Akt pathways. Collectively, our data demonstrated frequent overexpression and amplification PTP1B in gastric cancer, and further determined the oncogenic role of PTP1B in gastric carcinogenesis. Importantly, PTP1B amplification predicts poor survival of gastric cancer patients.
Insulin-like growth factor (IGF) signaling is involved in oral squamous cell carcinoma (OSCC), but IGF-1 receptor (IGF-1R)-mediated intricate regulatory networks among molecular interactions and signalling path ways in OSCC remain unclear. Here, we found that overexpression of IGF-1R and insulin receptor substrate-2 (IRS-2) was negatively associated with histological differentiation. IGF signaling stimulated OSCC cell growth. Conversely, overexpression of let-7b inhibited proliferation and colony formation and triggered S/G2 cell cycle arrest by targeting IGF-1R and IRS-2 through the Akt pathway. Also, the inverse relationship between expression of let-7b and IGF-1R/IRS-2 was confirmed in OSCC tumor xenografts and clinical specimens. Furthermore, by activating ERK1/2, IGF-1R transcriptionally upregulated IRS-2. Our results indicate that let-7b/IGF-1R-mediated crosstalk between IRS-2/Akt and MAPK is involved in OSCC and is a potential therapeutic target for therapy.
It has been documented in in vitro studies that zinc oxide nanoparticles (ZnO NPs) are capable of inducing oxidative stress, which plays a crucial role in ZnO NP-mediated apoptosis. However, the underlying molecular mechanism of apoptosis in neurocytes induced by ZnO NP exposure was not fully elucidated. In this study, we investigated the potential mechanisms of apoptosis provoked by ZnO NPs in cultured primary astrocytes by exploring the molecular signaling pathways triggered after ZnO NP exposure. ZnO NP exposure was found to reduce cell viability in MTT assays, increase lactate dehydrogenase (LDH) release, stimulate intracellular reactive oxygen species (ROS) generation, and elicit caspase-3 activation in a dose- and time-dependent manner. Apoptosis occurred after ZnO NP exposure as evidenced by nuclear condensation and poly(ADP-ribose) polymerase-1 (PARP) cleavage. A decrease in mitochondrial membrane potential (MMP) with a concomitant increase in the expression of Bax/Bcl-2 ratio suggested that the mitochondria also mediated the pathway involved in ZnO NP-induced apoptosis. In addition, exposure of the cultured cells to ZnO NPs led to phosphorylation of c-Jun N-terminal kinase (JNK), extracellular signal-related kinase (ERK), and p38 mitogen-activated protein kinase (p38 MAPK). Moreover, JNK inhibitor (SP600125) significantly reduced ZnO NP-induced cleaved PARP and cleaved caspase-3 expression, but not ERK inhibitor (U0126) or p38 MAPK inhibitor (SB203580), indicating that JNK signaling pathway is involved in ZnO NP-induced apoptosis in primary astrocytes.
The retinoblastoma protein (pRb) is required for cell-cycle exit of embryonic mammalian hair cells but is not required for hair cell fate determination and early differentiation, and this provides a strategy for hair cell regeneration by manipulating the pRb pathway. To reveal the mechanism of pRb functional modification in the inner ear, we compared the effects of attenuated pRb phosphorylation by an inhibitor of the Mitogen-Activated Protein (MAP) kinase pathway and an inhibitor of the Rb-Raf-1 interaction on cultured chicken otocysts. We demonstrated that the activity of pRb is correlated with its phosphorylation state, which is regulated by a newly established cell cycle-independent pathway mediated by the physical interaction between Raf-1 and pRb. The phosphorylation of pRb plays an important role during the early stage of inner ear development, and attenuated phosphorylation in progenitor cells leads to cell cycle arrest and increased apoptosis along with a global down-regulation of the genes involved in cell cycle progression. Our study provides novel routes to modulate pRb function for hair cell regeneration.
The expression and function of P-glycoprotein (P-gp) is associated with the phenotype of multi-drug resistance (MDR), leading chemotherapy failure of patients suffered with cancer. Grape seed procyanidin(GSP) is a natural polyphenol supplement with anti-inflammatory effect. Present study assessed a new use of GSP on the MDR reversal activity and its possible molecular mechanisms in MDR1-overpressing paclitaxel resistant ovarian cancer cells. Our results showed GSP significantly enhanced the cytotoxicity of paclitaxel and adriamycin in paclitaxel resistant A2780/T cells but its parental A2780 cells. Furthermore, GSP strongly inhibited P-gp expression by blocking MDR1 gene transcription, as well as, increased the intracellular accumulation of the P-gp substrate rhodamine-123 in A2780/T cells. Nuclear factor-κB(NF-κB) activity, IκB degradation level and NF-κB/p65 nuclear translocation induced by lipopolysaccharide (LPS) and receptor activator for nuclear factor-κB ligand (RANKL) were markedly inhibited by pre-treatment with GSP. Meanwhile, GSP inhibited MAPK/ERK pathway by decreasing the phosphorylation of ERK1/2, resulting in reduced the Y-box binding protein 1 (YB-1) activation with blocking its nuclear translocation. Moreover, the up-regulation of P-gp expression, the activation of AKT/NF-κB and MAPK/ERK pathway induced by LPS was attenuated by GSP administration. Compared with PDTC and U1026, inhibitor of NF-κB and MAPK/ERK respectively, GSP showed the same tendency of down-regulating NF-κB and MAPK/ERK mediated YB-1 activities. Thus, GSP reverses P-gp associated MDR by inhibiting the function and expression of P-gp through down-regulation of NF-κB activity and MAPK/ERK pathway mediated YB-1 nuclear translocation, offering insight into the mechanism of reversing MDR by natural polyphenol supplement compounds. GSP could be a new potential MDR reversal agent used for combination therapy with chemotherapeutics in clinic.
MicroRNAs (miRNAs) have been suggested to play a vital role in regulate tumor progression and invasion. However, the expression of miR-339-5p in colorectal cancer and its effects are not known. Here, we report that miR-339-5p is a tumor suppressor by regulating expression of PRL-1. In this study, we showed that downregulated miR-339-5p levels in colorectal cancer tissues and highly invasive CRC cell lines. Furthermore, enhancing the expression of miR-339-5p inhibited CRC cell growth, migration and invasion in vitro and suppressed tumor growth in vivo. We then screened and identified a novel miR-339-5p target, phosphatases of regenerating liver-1 1 (PRL-1), and it was further confirmed by luciferase assay. Overexpression of miR-339-5p would also reduce the expression of PRL-1 mRNA and protein. The reduced PRL-1 expression was associated with low expression of phosphorylated-extracellular signal-regulated kinase1/2 (p-ERK1/2). Conversely, reduction of miR-339-5p by inhibitors in cells stimulated these phenotypes. In conclusion, our results demonstrate that miR-339-5p functions as a tumor suppressor and plays a role in inhibiting growth and metastasis of CRC cells through targeting PRL-1 and regulating p-ERK1/2 .These findings suggest that miR-339-5p may be useful as a new potential therapeutic target for CRC.
Holothurian glycosaminoglycan (hGAG) is a high-molecular-weight form of fucosylated chondroitin sulfate and has an antithrombotic effect. Our previous studies demonstrated that hGAG efficiently inhibited tumor cell metastasis. The interplays between thrombosis and tumor progression may have a major impact on hematogenous metastasis. In this study, we demonstrated that the mouse melanoma B16F10 cells treated with hGAG displayed a significant reduction of metastasis and coagulation capacity in vitro and in vivo. Mechanistic studies revealed that hGAG treatment in B16F10 cells remarkably inhibited the formation of fibrin through attenuating the generation of activated Factor Xa (FXa), without affecting the expression of urokinase (uPA) and plasminogen activator inhibitor 1 (PAI-1) that involved in fibrinolysis. Moreover, hGAG treatment downregulated the transcription and protein expression of tissue factor (TF). Promoter deletions, site mutations and functional studies identified that the nuclear transcription factor NF-κB binding region is responsible for hGAG-induced inhibition of TF expression. While the hGAG treatment of B16F10 cells was unable to inhibit NF-κB expression and phosphorylation, hGAG significantly prevented nuclear translocation of NF-κB from the cytosol, a potential mechanism underlying the transcriptional suppression of TF. Moreover, hGAG markedly suppressed the activation of p38MAPK and ERK1/2 signaling pathways, the central regulators for the expression of metastasis-related matrix metalloproteinases (MMPs). Consequently, hGAG exerts a dual function in the inhibition of metastasis and coagulation activity in mouse melanoma B16F10 cells. Our studies suggest hGAG to be a promising therapeutic agent for metastatic cancer treatment.
BACKGROUND:
The dipeptidyl peptidase-4 inhibitor sitagliptin, a new anti-diabetic medicine, is effective in treating type 2 diabetes mellitus by increasing the activation and duration of action of glucagon-like peptide-1. Since atherosclerosis is the main pathological feature of diabetic cardiovascular complications, it is important to investigate the anti-atherosclerotic effect of sitagliptin and explore the relevant mechanisms.
METHODS:
Male apolipoprotein-E-knockout mice were randomly divided into two groups and fed either high-fat diet (HFD) or HFD plus sitagliptin at a concentration of 0.3% for 16 weeks. Body weight, food intake, blood glucose, serum lipids and adhesion molecules were measured. The atherosclerotic plaque area and its histological composition were analyzed using Sudan staining and immunohistochemistry. The expression of inflammatory cytokines (monocyte chemoattractant protein (MCP)-1 and interleukin (IL)-6) and the activation of AMP-activated protein kinase (AMPK) and mitogen-activated protein kinase (MAPK) in the aortas were determined using quantitative polymerase chain reaction and western blot, respectively.
RESULTS:
Mice treated with sitagliptin developed fewer atherosclerotic plaques than the control group (7.64 ± 1.98% vs 12.91 ± 1.15%, p < 0.001), particularly in the aortic arch and abdominal aorta, where plaques were decreased 1.92- and 2.74-fold, respectively (p < 0.05 and p < 0.01). Sitagliptin significantly reduced the content of collagen fiber in plaques 1.2-fold (p < 0.05). Moreover, sitagliptin significantly reduced the expression of monocyte chemoattractant protein-1 and interleukin-6 in the aorta (p < 0.01 and p < 0.05), as well as the serum levels of soluble vascular cell adhesion molecule-1 and P-selectin (both p < 0.05). In addition, Sitagliptin induced phosphorylation of AMPK and Akt (p < 0.05 and p < 0.01), while suppressed phosphorylation of p38 and extracellular signal-regulated kinase (Erk) 1/2 (p < 0.05 and p < 0.01) in aortas.
CONCLUSIONS:
Our present study indicates that sitagliptin can reduce the area of the atherosclerotic lesion, possibly by regulating the AMPK and MAPK pathways and then reducing leukocyte -endothelial cell interaction and inflammation reactions. These actions are independent of weight loss and glucose-reducing effects.
Successful placentation depends on the proper invasion of extravillous trophoblast (EVT) cells into maternal tissues. Previous reports have demonstrated that FoxM1 is oncogenic and plays important roles in angiogenesis, invasion, and metastasis. However, little is known about the roles of FoxM1 in the invasion of EVT cells. EGF, as a growth factor (epidermal growth factor), has been studied extensively in reproduction. JAR cells are a reliable model for studying early invasive trophoblast regulation. We have observed the relationship between EGF and FoxM1 in JAR cells by using specific inhibitors for the intervention in and study of potential signal pathways. We have also tested the ability of JAR cells to be influenced by the expression of FoxM1. Our data indicate that EGF promotes FoxM1 expression through the ERK signal pathway. Over-FoxM1 expression upregulates the ability of JAR cells to migrate and invade and vice versa. Our investigation of FoxM1 should provide new insights into the molecular mechanisms of EVT invasion.
The effect of the total saponins from Rosa laevigata Michx fruit (RLTS) against acetaminophen (APAP)-induced liver damage in mice was evaluated in the present paper. The results showed that RLTS markedly improved the levels of liver SOD, CAT, GSH, GSH-Px, MDA, NO and iNOS, and the activities of serum ALT and AST caused by APAP. Further research confirmed that RLTS prevented fragmentation of DNA and mitochondrial ultrastructural alterations based on TdT-mediated dUTP nick end labeling (TUNEL) and transmission electron microscopy (TEM) assays. In addition, RLTS decreased the gene or protein expressions of cytochrome P450 (CYP2E1), pro-inflammatory mediators (IL-1β, IL-4, IL-6, TNF-α, iNOS, Bax, HMGB-1 and COX-2), pro-inflammatory transcription factors (NF-κB and AP-1), pro-apoptotic proteins (cytochrome C, p53, caspase-3, caspase-9, p-JNK, p-p38 and p-ERK), and increased the protein expressions of Bcl-2 and Bcl-xL. Moreover, the gene expression of IL-10, and the proteins including LC3, Beclin-1 and Atg5 induced by APAP were even more augmented by the extract. These results demonstrate that RLTS has hepatoprotective effects through antioxidative action, induction of autophagy, and suppression of inflammation and apoptosis, and could be developed as a potential candidate to treat APAP-induced liver damage in the future.
Triple-negative breast cancer (TNBC) is an aggressive breast cancer with a generally poor prognosis. Due to lack of specific targets for its treatment, an efficient therapy is needed. G protein-coupled estrogen receptor (GPER), a novel estrogen receptor, has been reported to be expressed in TNBC tissues. In this study, we investigated the effects of blocking non-genomic signaling mediated by the estrogen/GPER pathway on cell viability and motility in the TNBC cells. GPER was strongly expressed in the TNBC cell lines MDA-MB-468 and MDA-MB-436, and the estrogen-mediated non-genomic ERK signaling activated by GPER was involved in cell viability and motility of TNBC cells. Treatment with 17β-estradiol (E2), the GPER-specific agonist G-1 and tamoxifen (TAM) led to rapid activation of p-ERK1/2, but not p-Akt. Moreover, estrogen/GPER/ERK signaling was involved in increasing cell growth, survival, and migration/invasion by upregulating expression of cyclinA, cyclinD1, Bcl-2, and c-fos associated with the cell cycle, proliferation, and apoptosis. Immunohistochemical analysis of TNBC specimens showed a significantly different staining of p-ERK1/2 between GPER-positive tissues (58/66, 87.9%) and GPER-negative tissues (13/30, 43.3%). The positivity of GPER and p-ERK1/2 displayed a strong association with large tumor size and poor clinical stage, indicating that GPER/ERK signaling might also contribute to tumor progression in TNBC patients which corresponded with in vitro experimental data. Our findings suggest that inhibition of estrogen/GPER/ERK signaling represents a novel targeted therapy in TNBC.
Matrix metalloproteinases (MMP) play a pivotal role in the pathogenesis of cardiovascular diseases. Their expressions are altered in response to a variety of stimuli, including growth factors, inflammatory markers, and cytokines. In this study, we demonstrated that platelet-derived growth factor-BB (PDGF-BB) induces a dose- and time-dependent increase in MMP-2 expression in rat vascular smooth muscle cells (VSMC). Treatment with either the Rho-associated protein kinase (ROCK) inhibitor Y-27632 or suppression of ROCK-1/2 by small interfering RNA technology significantly reduced the MMP-2 expression, thus suggesting that ROCK regulates such expression. Similar results were observed when VSMC were pretreated with either U0126 or SB203580, which are selective inhibitors of extracellular signal-regulated kinase and p38 mitogen-activated protein kinase, respectively, thus suggesting that these kinases are important for the induction of MMP-2 expression by PDGF-BB. In conclusion, these results described a novel mechanism in atherosclerosis through PDGF-BB signaling in VSMC, in which MMP-2 expression is induced via extracellular signal-regulated kinases and p38 mitogen-activated protein kinase phosphorylation, as well as ROCK.
Exposure to elevated levels of fluoride can cause a variety of adverse effects in fish. Previously we showed that fluoride causes injuries and apoptosis in the gills of Cyprinus carpio. In this study, the effects of fluoride on caspase-3 activity and on accumulation of proteins in the MAPKs pathways were evaluated using Western blotting and immunohistochemistry methods in vivo and in vitro. In vivo experiments showed that the caspase-3 activity increased with fluoride exposure level in a dose-dependent pattern Western blotting and immunohistochemistry results indicated that ERK relative activation tended to decrease as a function of fluoride exposure concentration. In contrast, relative activation of JNK increased with fluoride exposure level. Fluoride exposure did not appear to affect p38 activation. Furthermore, pretreatment of branchial cells with MAPK-specific inhibitors effectively prevented JNK induction and ERK inhibition, respectively, as well as reversed caspase-3 activity in fluoride-treated branchial cells. Our results indicate that activation of JNK and inactivation of ERK were caused by increased ROS and decreased antioxidant capacity in the gills of chronically exposed C. carpio described previously, which eventually caused the observed apoptosis in the fluoride-exposed gills and cells in C. carpio. JNK activation and ERK inactivation mechanism play a crucial role in gill impairment induced by chronic fluorosis. These findings contribute to a better understanding of the initial molecular and cellular events in the gill of fish chronically exposed to fluoride.
The aim of this study was to investigate whether the mitochondrial permeability transition pore (MPTP) opening was involved in the protective effects of CB2 receptor against ischemia-reperfusion (I-R) injury. For this, isolated perfused rat hearts were subjected to 30 min global ischemia followed by 120 min reperfusion, and left ventricle function was recorded. At the end of reperfusion, the infarct size in the hearts was measured by staining with triphenyltetrazolium chloride. MPTP opening and the mitochondrial membrane potential (ΔΨ(m)) were measured by flow cytometry. Western blot analysis of cytochrome c in the mitochondrion and cytosol, as well as ERK1/2 and p-ERK1/2 were performed. Administration of CB2 receptor agonist JWH133 before ischemia significantly improved the recovery of cardiac ventricular function during reperfusion, increased coronary flow, reduced infarct size, prevented the loss of ΔΨ(m) and MPTP opening, reduced the release of cytochrome c from mitochondria, and increased levels of p-ERK1/2. These effects of JWH133 were abolished by pretreatment with CB2 receptor antagonist AM630, or ERK1/2 inhibitor PD98059. Furthermore, JWH133 reversed the MPTP opening induced by atractyloside. The protective effect of JWH133 on the heart against I-R injury may be through increased ERK1/2 phosphorylation, inhibiting MPTP opening.
Dentin, the predominant mineralized tissue of the tooth, comprises an extracellular matrix of collagen and a heterogeneous mixture of non-collagenous components, many of which have cellular signaling properties. These properties may be important in signaling stem cell involvement in tissue regeneration following injury and the present study investigates their morphogenic effects on differentiation of Bone Marrow Stromal Stem Cells (BMMSCs) in vitro. Non-collagenous dentin matrix proteins (DMPs) were isolated from healthy human teeth and their effects on BMMSCs behavior examined during in vitro culture. In vitro, DMPs enhanced alkaline phosphatase activity and mineralization in BMMSCs cultures as well as increasing the expression of dentinogenic and osteogenic differentiation markers (including runt-related transcription factor 2, osterix, bone sialoprotein, dentin sialophosphoprotein and osteocalcin) at both transcript and protein levels, with 10 μg/mL DMPs being the optimal stimulatory concentration. Expression of phosphor-ERK/phosphor-P38 in BMMSCs was up-regulated by DMPs and, in the presence of the ERK1/2- and p38-specific inhibitors, the differentiation of BMMSCs was inhibited. These data indicate that DMPs promote the dentinogenic/osteogenic differentiation of BMMSCs via the ERK/p38 MAPK pathways.
Angiogenesis is a process that forms new blood capillaries from existing vessels, which is of great physiological and pathological significance. Although recent studies provide evidence that cuprous oxide nanoparticles (CO-NPs) may have biomedical potential, the mechanisms of CO-NPs in angiogenesis have not been investigated to date. We have studied the anti-angiogenic properties of CO-NPs on primary human umbilical vein endothelial cells (HUVECs). We found that CO-NPs were able to induce cell morphology changes and suppress cell proliferation, migration and tube formation in vitro and in vivo dose dependently. Furthermore, CO-NPs could induce cell apoptosis both at the early and late apoptotic stage and induce cell cycle arrest at S phase in a dose dependent manner. As signalling via the vascular endothelial growth factor receptor-2 (VEGFR2) is critical for angiogenic responses, we further explored the expression of VEGFR2 after the treatment of CO-NPs. They were found to inhibit VEGFR2 expression dose and time dependently both at the protein and mRNA level while had no effect on VEGF and VEGFR1 expression. Together, we report for the first time that CO-NPs can act as an anti-angiogenic agent by suppressing VEGFR2 expression, which may be a potential nanomedicine for angiogenesis therapy.
Sertoli cells play a pivotal role in supporting proliferation of germ cells and differentiation during spermatogenesis in mammals. Nanomolar concentrations of Bisphenol A (BPA) can significantly stimulate the proliferation of mouse immature Sertoli (TM4) cells. However, mechanisms by which BPA caused these effects were still unclear. In the present study, an inverse U-shaped curve was observed when treating TM4 cells with increasing doses of BPA: 1 to 10nM BPA significantly stimulated the proliferation of TM4 cells and increased the proportion of cells in S phase; >1 μM BPA caused lesser proliferation of cells. Exposure of TM4 cells to G15 or ICI 182,780, which are specific antagonists of GPR30 and estrogen receptor α/β (ERα/β), respectively, abolished BPA-induced proliferation of cells, which suggests that both GPR30 and ERα/β were involved in the observed effects of BPA. Furthermore, exposure to BPA caused rapid (5 min) activation of ERK1/2 via both GPR30 and ERα/β. Blocking the GPR30/EGFR signal transduction pathway by antagonists suppressed both phosphorylation of ERK and BPA-induced cell proliferation. BPA up-regulated mRNA and protein expression of GPR30 in a concentration-dependent manner. In summary, the results reported here indicated that activating ERK1/2 through GPR30 and ERα/β is involved in low doses of BPA that promoted growth of Sertoli TM4 cells. The GPR30/EGFR/ERK signal is the downstream transduction pathway in BPA-induced proliferation of TM4 Sertoli cells.
The present study was to investigate the effects and possible mechanisms of the total saponins from Dioscorea nipponica Makino (TSDN) against CCl₄-induced hepato-toxicity in mice. The mice were orally administrated with TSDN for seven days and then given CCl₄ (0.3%, 10 ml/kg i.p.). The results showed that TSDN significantly attenuated the activities of ALT and AST, consistent with hematoxylin-eosin staining. The ALP levels and relative liver weight were significantly decreased by TSDN compared with model group. Moreover, TSDN dramatically decreased MDA, iNOS and NO levels, while the levels of GSH, GSH-Px and SOD were increased. Further investigations showed that TSDN inhibited CCl₄-induced metabolic activation and CYP2E1 expression, down-regulated the levels of MAPKs phosphorylation, NF-κB, HMGB1, COX-2 as well as effectively suppressed the expressions of Caspase-3, Caspase-9, PARP and Bak. Quantitative real-time PCR assay demonstrated that TSDN obviously decreased the gene expressions of TNF-a, IL-1β, IL-6, IL-10, Fas, FasL, Bax as well as modulated Bcl-2 mRNA level. This is the first time to report the protective actions of the TSDN against CCl₄-induced liver damage in mice through suppression of inflammation and apoptosis. This natural product should be developed as a new drug for treatment of liver injury in future.
This study was conducted to demonstrate myocardial protective effects and possible underlying mechanisms of vitexin on myocardial ischemia/reperfusion (I/R) injury in rats. Occluding the anterior descending artery for 30 min and restoring blood perfusion for 60 min in rat established a model of myocardial I/R. The elevation of the ST segment of Electrocardiograph (ECG) was observed. The infarct size of the rat heart was assessed by triphenyltetrazolium chloride staining (TTC). LDH, CK, SOD activities and MDA content were determined. An immunohistochemical analysis was applied to measure the expression of myocardial NF-κBp65 and TNF-α. ERK/phospho-ERKand c-Jun/phospho-c-Jun protein expression was examined via Western Blot. Vitexin significantly reduced the elevation of the ST segment of ECG and myocardial infarct size. LDH and CK activities and MDA content were attenuated in serum, while SOD activity was markedly enhanced. Vitexin significantly attenuated I/R-induced increases of myocardial NF-κB and TNF-α. Moreover, Western Blot analysis presented that vitexin markedly enhanced the expression of phospho-ERK and weakened the expression of phospho-c-Jun compared to I/R group. The significant protective effect against myocardial ischemical/reperfusion injury in rat, which is exhibited by vitexin, may be related to its antioxidative and anti-inflammatory effects by regulating inflammatory cytokines and the MAPK pathway.
Oocytes with germinal vesicles (GVs) replaced with somatic nuclei exhibit meiotic abnormalities. Although this suggests an exclusive role for GV material in meiosis, mechanisms by which a lack of GV material causes meiotic defects are unknown. Knowledge of these mechanisms will help us to understand meiotic control, nuclear-cytoplasmic interactions, and cellular reprogramming. This study showed that although oocytes with prometaphase I chromosomes replaced with primary spermatocyte nuclei (PSN) did not, oocytes with GV replaced with PSN (PSG oocytes) did display meiotic defects. Among the defects, insufficient chromosome condensation with chromosome bridges was associated with spindle abnormalities. Abnormal spindle migration, cortical nonpolarization, and the aberrant spindle caused randomly positioning of cleavage furrows, leading to large first polar bodies (PB1) and unequal allocation of chromosomes and mitogen-activated protein kinases (MAPK) between oocyte and PB1. Spindle assembly checkpoint was activated but did not stop the incorrect division. The unequal MAPK allocation resulted in differences in pronuclear formation and PB1 degeneration; oocytes receiving more MAPK were more capable of forming pronuclear rudiments, whereas PB1 receiving more MAPK degenerated sooner than those that received less. Because none of the PSG oocytes or the enucleated GV oocytes injected with sperm heads showed cortical polarization in spite of chromosome localization close to the oolemma and because the PSG oocytes receiving more MAPK could form only pronuclear rudiments and not normal pronuclei, we suggest that the GV material plays essential roles in polarization and pronuclear formation on top of those played by chromosomes or MAPK. In conclusion, using PSG oocytes as models, this study has revealed the primary pathways by which a lack of GV material cause meiotic defects, laying a foundation for future research on the role of GV material in oocyte meiotic control.
Hyperuricaemia is a disorder of purine metabolism, and is strongly associated with insulin resistance and abnormal glucose metabolism. As the producer of insulin, pancreatic β cells might be affected by elevated serum uric acid levels and contribute to the disregulated glucose metabolism. In this study, we investigated the effect of high uric acid on rat pancreatic β cell function. Under high uric acid condition, proliferation of pancreatic β cells was inhibited, production of reactive oxygen species increased, and glucose stimulated insulin secretion was also compromised. Further examination on signal transduction pathways revealed that uric acid-induced ROS is involved in the activation of adenosine monophosphate-activated protein kinase (AMPK), and extracellular signal-regulated kinase (ERK). Pharmacological inhibition of ERK activation rescued β cells from growth inhibition. More importantly, activation of ERK induced by uric acid is significantly diminished by AMPK inhibitor, indicating ERK as a downstream target of AMPK in response to high uric acid condition. We also investigated the transportation channel for uric acid into pancreatic β cells. While major urate transporter URAT1 is not expressed in β cells, organic anion transporter (OAT) inhibitor successfully blocked the activation of ERK by uric acid. Our data indicate that high uric acid levels induce oxidative damage and inhibit growth of rat pancreatic β cells by activating the AMPK and ERK signal pathways. Hyperuricemia may contribute to abnormal glucose metabolism by causing oxidative damage and function inhibition of pancreatic β cells.
Pulmonary fibrosis is a serious and irreversible lung injury with obscure etiologic mechanisms and no effective treatment to date. This study explored a crucial link between oxidative stress and pulmonary fibrogenesis, focusing on nuclear factor erythroid 2-related factor 2 (Nrf2), a core transcription factor in antioxidative regulation systems. Treatment of C57 BL/6 mice with bleomycin increased fibroblast viability and collagen production and significantly downregulated Nrf2. In addition, prominent oxidative stress was indicated by changes in superoxide dismutase, catalase activity, and glutathione and thiobarbituric acid-reactive substance levels. In a cell-based model, bleomycin suppressed Nrf2 activation via extracellular signal-related kinase phosphorylation, enhancing intracellular reactive oxygen species in lung fibroblasts and stimulating abnormal cell proliferation and collagen secretion. To confirm this novel mechanism of bleomycin-induced fibrogenesis, we attempted to upregulate Nrf2 and related antioxidant proteins in bleomycin-treated fibroblasts using a putative Nrf2 activator, caffeic acid phenethyl ester, and the results showed that bleomycin-induced fibroblast proliferation and collagen content were attenuated through improved redox balance. Collectively, these results disclose a potential regulatory mechanism in pulmonary fibrosis that will aid the development of new therapies.
Tropomyosin-related kinase A (TrkA) is a receptor-type protein tyrosine kinase and exploits pleiotypic roles via nerve growth factor (NGF)-dependent or NGF-independent mechanisms in various cell types. Here, we showed that the inhibition of TrkA activity by GW441756 resulted in the suppression of tyrosine phosphorylation of cellular proteins including extracellular signal-regulated protein kinase (ERK) and c-Jun N-terminal kinase (JNK). To find novel targets associated with TrkA-mediated tyrosine phosphorylation signaling pathways, we investigated GW441756 effects on TrkA-dependent targets in SK-N-MC neuroblastoma cells by proteomic analysis. The major TrkA-dependent protein spots controlled by GW441756 were determined by PDQuest image analysis, identified by MALDI-TOF MS and MALDI-TOF/TOF MS/MS, and verified by 2DE/Western blot analysis. Thus, we found that most of the identified protein spots were modified forms in a normal condition, and their modifications were regulated by TrkA activity. Especially, our results demonstrated that the modifications of α-tubulin and heterogeneous nuclear ribonucleoproteins C1/C2 (hnRNP C1/C2) were significantly upregulated by TrkA, whereas α-enolase modification was downregulated by TrkA, and it was suppressed by GW441756, indicating that TrkA activity is required for their modifications. Taken together, we suggest here that the major novel TrkA-dependent targets such as α-tubulin, hnRNP C1/C2, and α-enolase could play an essential role in TrkA-mediated tyrosine phosphorylation signaling pathways via regulation of their posttranslational modifications.
CD133 is widely expressed in colorectal cancer (CRC) tissues and cell lines. This protein has been used as a marker of CRC cancer stem cells, although the function and mechanism of CD133 in CRC invasion and metastasis remain unclear. In our study, we examined the role of CD133 in CRC invasion in vitro and investigated the mechanism involved in CD133-related invasion. CD133(high) and CD133(low) HCT116 cells were isolated, and the proliferation and invasive ability of these two subpopulations were tested. CD133(high) HCT116 cells exhibited greater proliferation and invasion compared with CD133(low) HCT116 cells. CD133 knockdown (using CD133 small-interfering [si]RNA) inhibited the invasive activity of CD133si-HCT116 cells. For the first time, we found that the expression of tissue inhibitor of matrix metalloproteinases-2 (TIMP-2) was down-regulated in CD133si-HCT116 cells. In addition, for the TIMP-2si-HCT116 cells (transfected with TIMP-2 siRNA), in vitro invasion was significantly decreased, whereas the expression of CD133 remained unchanged. Finally, the metalloproteinase 2 and MEK/ERK signaling pathways were examined, and no significant change was observed after the knockdown of CD133 or TIMP-2 in HCT116 cells. In conclusion, we demonstrated that CD133 plays an important role in HCT116 cell invasion, and for the first time, we found that CD133 knockdown significantly down-regulated TIMP-2 expression, which suggests that CD133 affects the invasive ability of HCT116 cells by regulating TIMP-2.
Endocrine therapy is an important therapeutic approach for the treatment of oestrogen receptor (ER)-positive breast cancer. However, a number of these endocrine therapies can fail when the tumour loses its ER expression during treatment. To date, few studies have explored the potential clinical significance of traditional Chinese medicine in inducing the reversal of resistance to endocrine therapy in breast cancers. We used the ERα-negative MCF7 breast cancer cell line to create a tamoxifen (TAM)-resistant cell line, MCF7/TAM cells. After treating MCF7/TAM cells with ELE to induce the re-expression of ERα, we investigated the role and molecular mechanisms by which elemene (ELE) promotes the reversal of resistance to endocrine therapy. We discovered that treatment with 10 μg/ml ELE restored the sensitivity of MCF7/TAM cells to TAM. RT-PCR analysis revealed that ELE treatment upregulated ERα mRNA levels in MCF7/TAM cells, and immunohistochemistry confirmed the upregulation of ERα expression. Western blot analysis revealed that ELE treatment decreased the protein expression levels of Ras, MEK1/2 and p-ERK1/2 in MCF7/TAM cells. The loss of ERα expression was the primary reason for TAM resistance in MCF7 cells. The ELE-induced reversal of TAM resistance was mediated by the upregulation of ERα mRNA and the re-expression of ERα through the MAPK pathway.
Insulin-like growth factor 1 (IGF-1) is a potent mitogenic protein which can enhance the osteogenic differentiation of periodontal ligament (PDL) fibroblasts. However, it remains unclear whether IGF-1 can stimulate the osteogenic differentiation and osteogenesis of human periodontal ligament stem cells (PDLSCs). In this study, STRO-1(+) PDLSCs were isolated from human PDL tissues, treated with IGF-1, and their osteogenic capacity was investigated in vitro and in vivo. Dimethyl-thiazol-diphenyl tetrazolium bromide assay and flow cytometry results demonstrated that 10-200 ng/mL IGF-1 can stimulate the proliferation ability of PDLSCs and 100 ng/mL is the optimal concentration. Exogenous IGF-1 can modify the ultrastructure, enhance the alkaline phosphatase activity, the mineralization ability of PDLSCs, and increase the expression of osteogenic markers (runt-related transcription factor 2, osterix, and osteocalcin) at mRNA and protein levels. In vivo transplantation illustrated that IGF-1 treated implants generated more mineralized tissues, and presented stronger expression of RUNX2, OSX, and OCN than control group. Moreover, the expression of phosphor-ERK and phosphor-JNK in these stem cells was upregulated by IGF-1, indicating that MAPK signaling pathway was activated during the osteogenic differentiation of PDLSCs mediated by IGF-1. Together, the results showed that IGF-1 can promote the osteogenic differentiation and osteogenesis of STRO-1(+) PDLSCs via ERK and JNK MAPK pathway, suggesting that IGF-1 is a potent agent for stem cell-based periodontal tissue regeneration.
We examined the effects of anti-six-transmembrane epithelial antigen of the prostate-4 (STEAP4) antibodies on glucose transport in mature adipocytes and determined the mechanism of insulin resistance in obesity. Western blotting was performed to determine STEAP4 expression, to assess translocation of insulin-sensitive glucose transporter 4 (GLUT4), and to measure phosphorylation and total protein content of insulin-signaling proteins. Confocal laser microscopy and flow cytometry were used to detect intracellular reactive oxygen species (ROS) and fluctuations in mitochondrial membrane potential (ΔΨ). ATP production was measured by using a luciferase-based luminescence assay kit. After the application of anti-STEAP4 antibodies at 0.002 mg/mL, adipocytes exhibited reduced insulin-stimulated glucose transport by attenuating the phosphorylation of IRS-1, PI3K (p85), and Akt. The antibodies also potentially increase the level of ROS and decrease cellular ATP production and ΔΨ. In conclusion, (i) STEAP4 regulates the function of IRS-1, PI3K, and Akt and decreases insulin-induced GLUT4 translocation and glucose uptake; (ii) ROS-related mitochondrial dysfunction may be related to a reduced IRS-1 correlation with the PI3K signaling pathway, leading to insulin resistance. These observations highlight the potential role of STEAP4 in glucose homeostasis and possibly in the pathophysiology of type 2 diabetes related to obesity and may provide new insights into the mechanisms of insulin resistance in obesity.
Oxidative stress is a major cause in neurodegenerative diseases including Alzheimer's disease (AD), Parkinson's disease (PD), and cerebral ischemia. Ginsenoside Rg1, a natural product extracted from Panax ginseng C.A. Meyer, has been reported to exert notable neuroprotective activities, which partly ascribed to its antioxidative activity. However, its molecular mechanism against oxidative stress induced by exogenous hydrogen peroxide (H(2)O(2)) remained unclear. In this study, we investigated its effect on H(2)O(2)-induced cell death and explored possible signaling pathway in PC12 cells. We proved that pretreatment with Rg1 at concentrations of 0.1-10 μM remarkably reduced the cytotoxicity induced by 400 μM of H(2)O(2) in PC12 cells by MTT and Hoechst and PI double staining assay. Of note, we demonstrated the activation of NF-κB signaling pathway induced by H(2)O(2) thoroughly in PC12 cells, and Rg1 suppressed phosphorylation and nuclear translocation of NF-κB/p65, phosphorylation and degradation of inhibitor protein of κB (IκB) as well as the phosphorylation of IκB-kinase complex (IKK) by western blotting or indirect immunofluorescence assay. Besides, Rg1 also inhibited the activation of Akt and the extracellular signal-regulated kinase 1/2 (ERK1/2). Furthermore, the protection of Rg1 on H(2)O(2)-injured PC12 cells was attenuated by pretreatment with two NF-κB pathway inhibitors (JSH-23 or BOT-64). In conclusion, our results suggest that Rg1 could rescue the cell injury by H(2)O(2) via down-regulation NF-κB signaling pathway as well as Akt and ERK1/2 activation, which put new evidence on the neuroprotective mechanism of Rg1 against the oxidative stress and the regulatory role of H(2)O(2) in NF-κB pathway in PC12 cells.
INTRODUCTION:
Since the discovery of the (pro)renin receptor (PRR), it has been considered as a novel bioactive molecule of the renin-angiotensin system (RAS). The activation of PRR can elicit a series of angiotensin II (AngII)-independent effects.
MATERIALS AND METHODS:
In this study, we investigated the effects of prorenin and PRR on the proliferation of human umbilical artery smooth muscle (HUASM) cells and explored the possible mechanisms underlying these effects.
RESULTS:
The binding of prorenin to PRR can promote proliferation and upregulate the anti-apoptotic protein Bcl-2 and downregulate the pro-apoptotic protein Bax independently of AngII in HUASM cells. In addition, the binding of prorenin to PRR can also increase the production of reactive oxygen species (ROS) and the phosphorylation of extracellular signal-regulated kinase (ERK1/2) independently of AngII. The pretreatment of HUASM cells with an NADPH oxidase inhibitor DPI decreased the production of ROS and also decreased the phosphorylation of ERK1/2. Furthermore, pretreatment of HUASM cells with DPI and the ERK1/2 inhibitor PD98059 significantly attenuated the prorenin-induced proliferation and regulation of apoptosis factors.
CONCLUSION:
Binding of prorenin to PRR can induce HUASM cell proliferation via the ROS generation and ERK1/2 activation.
© The Author(s) 2014.
BACKGROUND:
Recently, research has indicated that leptin plays a protective role in traumatic brain and liver injury. We studied the protective effect of leptin on intestinal I/R injury and examined its mechanism by using mice intestinal I/R model and murine peritoneal macrophage hypoxia/reoxygenation (H/R) injury model.
METHODS:
Leptin was intraperitoneally administrated at 45 minutes after ischemia, then reperfusion for two hours. Cells were treated with different concentrations of leptin at three hours after hypoxia, then reoxygenation for six hours. Mice intestines were harvested for histopathologic properties. The malondialdehyde, nitric oxide (NO), interleukin-6, and total antioxidative capacity were detected according to respective assay kit. Phosphorylated extracellular regulated kinase1/2 (p-ERK1/2) and phosphorylated cytosolic phospholipase A(2) (p-cPLA2) were determined by Western blot assay.
RESULTS:
Here, we show that leptin reduced intestinal histologic alterations, malondialdehyde and interleukin-6 levels but increased the endogenous leptin expression and NO production in the intestines. Leptin also increased the NO and total antioxidative capacity levels in cells. We further demonstrated that leptin markedly activated ERK1/2 in the intestines and activated ERK1/2 and cPLA2 in the cells. Moreover, the protective effect of leptin against intestinal I/R injury and elevated NO production was attenuated by blocking the ERK1/2 pathway.
CONCLUSIONS:
These data demonstrate that leptin ameliorated intestinal I/R and peritoneal macrophage H/R injury by enhancing ERK1/2 phosphorylation and promoting the NO production signaling pathway.
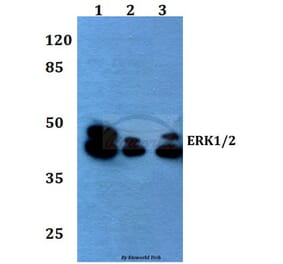
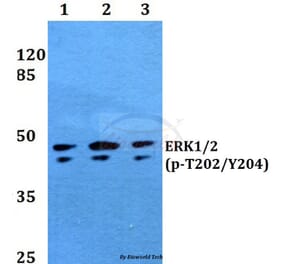

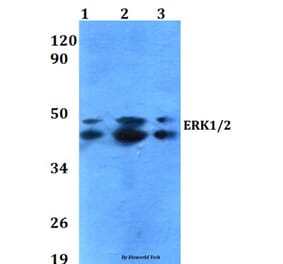
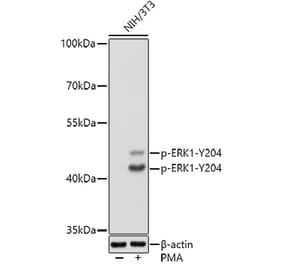
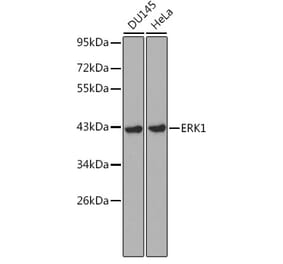
![Western Blot - Anti-ERK1 Antibody [ARC2591] (A306776) - Antibodies.com](https://cdn.antibodies.com/image/catalog/306/A306776_1.jpg?profile=product_alternative)
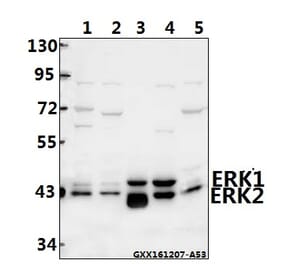






![Western Blot - Anti-ERK1 Antibody [ARC2591] (A306776) - Antibodies.com](https://cdn.antibodies.com/image/catalog/306/A306776_1.jpg?profile=product_alternative)