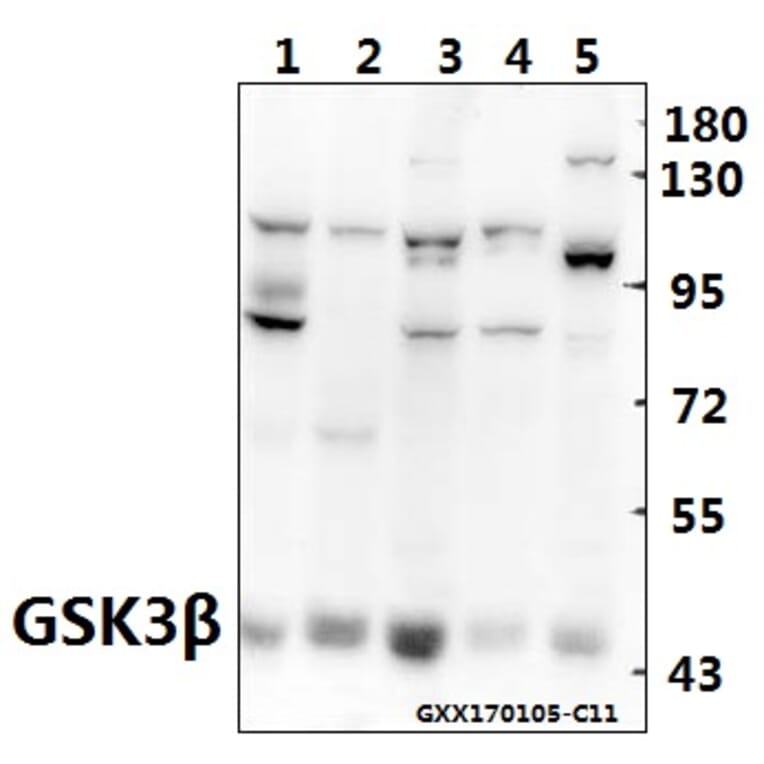
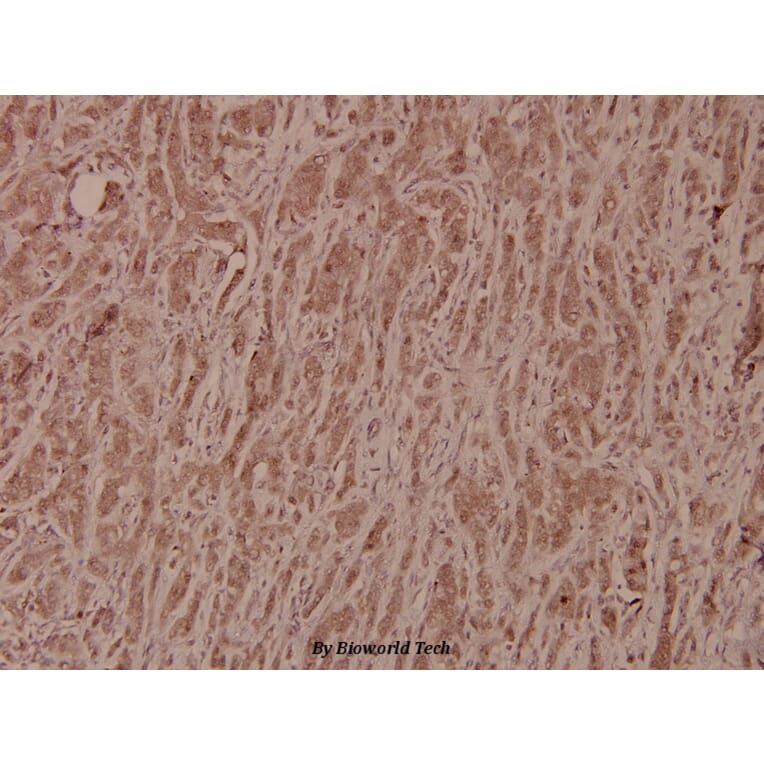
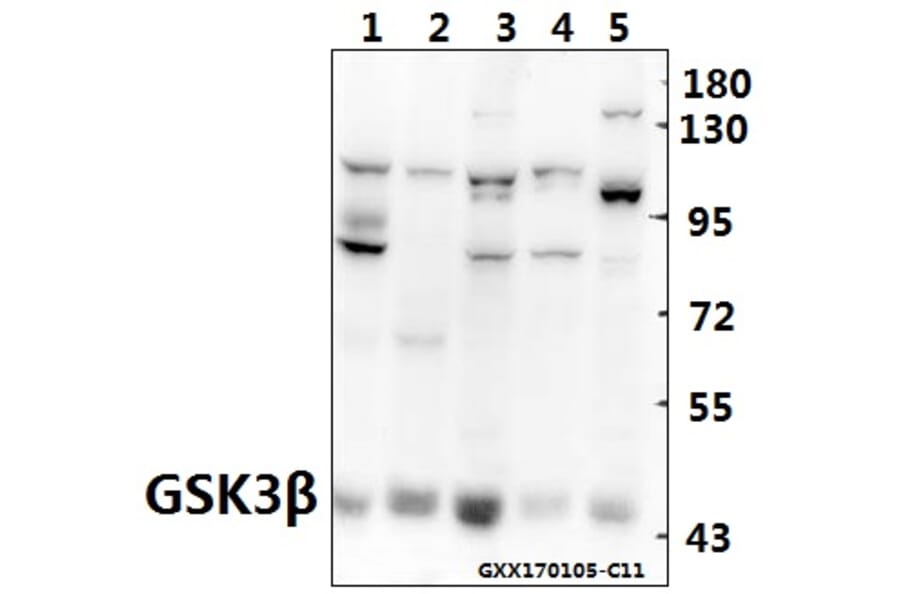
Unconjugated
Fangchinoline, an important compound in Stephania tetrandra S. Moore, as a novel antitumor agent, has been implicated in several types of cancers cells except gastric cancer. To investigate whether fangchinoline affects gastric cancer cells, we detected the signaling pathway by which fangchinoline plays a role in different human gastric cancer cells lines. We found that fangchinoline effectively suppressed proliferation and invasion of SGC7901 cell lines, but not MKN45 cell lines by inhibiting the expression of PI3K and its downstream pathway. All of the Akt/MMP2/MMP9 pathway, Akt/Bad pathway, and Akt/Gsk3β/CDK2 pathway could be inhibited by fangchinoline through inhibition of PI3K. Taken together, these results suggest that fangchinoline targets PI3K in tumor cells that express PI3K abundantly and inhibits the growth and invasive ability of the tumor cells.
Epithelial-mesenchymal transition (EMT) plays an important role in tumor metastasis of human nonsmall cell lung cancer (NSCLC). The Wnt pathway is identified as a key regulator of normal tissue development, and its aberrant activation contributes to the process of EMT. The secreted frizzled-related protein 1 (sFRP1), a Wnt-signaling antagonist, is downregulated in many tumors, including lung cancer. However, the role of sFRP1 in EMT and tumor metastasis remains unclear. In this study, we found that sFRP1 was dramatically downregulated in transforming growth factor β1 (TGF-β1)-induced EMT in the A549 human lung cancer cell line. Restoration of sFRP1 could inhibit the TGF-β1-induced EMT phenotype and tumor metastasis of the A549 cell line both in vitro and in vivo through inhibition of the Wnt pathway. Furthermore, FH535, a reversible Wnt-signaling inhibitor, exerted a similar effect on the TGF-β1-induced EMT phenotype. These results indicate that sFRP1, an endogenous antagonist of the Wnt pathway, inhibits TGF-β1-induced EMT, and might be a potential biomarker for the treatment of NSCLC.
Prolonged ingestion of a cholesterol-enriched diet induces chronic, auto-inflammatory responses resulting in significant health problems including colorectal cancer. Inflammasomes are thought to mediate intestinal homeostasis, and their dysregulation contributes to inflammatory bowel diseases and colitis-associated cancer (CAC). However, in vitro and in vivo information regarding the inflammation-inducing and tumor-promoting effect of cholesterol is lacking. Here we show that the cholesterol promoted colon carcinogenesis in azoxymethane (AOM)-treated mice through activating the NLRP3 inflammasome. High cholesterol diet (HCD) significantly increased inflammatory responses and tumor burden. Cholesterol crystals, detected in the colon of mice fed with HCD, also promoted NLRP3 inflammasome activation in macrophages, as indicated by elevated expression of cleaved caspase-1, formation of NLRP3-ASC-caspase-1 complex assembly, and higher IL-1β secretion. Importantly, cholesterol was found to inhibit the activity of AMPKα in macrophages, leading to a significant production of mitochondrial ROS, which in turn activated the NLRP3 inflammasome. Moreover, crystal uptake and cathepsin B accounted for cholesterol crystal-induced inactivation of AMPKα. Finally, HCD-induced increase in IL-1β secretion, macrophage infiltration and tumor burden was diminished by the deletion of NLRP3 in AOM-treated mice. Taken together, our findings demonstrate that the pro-inflammatory and cancer-promoting effects of HCD are mediated by the activation of NLRP3 inflammasome. Our study extended our knowledge on how dietary choices can influence processes involved in chronic inflammatory disorders and colorectal cancer.
Snail is closely linked to tumor invasion, metastasis, and recurrence and indicates prognosis of patients suffering from cancer. Overexpression of Snail increases motility and invasiveness of cancer cells, which has become target for anti-metastatic treatment. Oroxylin A, a natural compound extracted from Scutellaria radix, has been reported to inhibit invasion and migration in breast cancer. In this study, we investigated the anti-invasive effect of oroxylin A on lung cells and uncovered its underlying mechanism. The results suggested that oroxylin A could inhibit migration and invasion in Snail-expressing 95-D, and A549 cells whereas it had little effect on non-expressing GLC-82 cells. Furthermore, enhanced Snail expression after transfection of Snail vector in GLC-82 cells is decreased by oroxylin A. Snail can also induce epithelial-mesenchymal transition. We found oroxylin A could reverse TGFβ1-induced epithelial-mesenchymal transition by inhibiting Snail expression. As a result, oroxylin A up-regulated E-cadherin expression and down-regulated vimentin, MMP-9, and CD44v6 expression, which could lead to the inhibition of tumor migration and invasion. Mechanically, we demonstrated that oroxylin A suppressed activation of ERK instead of AKT pathway and then promoted activation of GSK-3β to reduce Snail protein content. Finally, we established transplanted, metastatic, and orthotopic models of A549 cells, and found that oroxylin A inhibited the growth and lung metastasis of A549 cells in vivo. Taken together, we proposed that oroxylin A might be a promising candidate targeting tumor metastasis.
© 2016 Wiley Periodicals, Inc.
Hepatocellular carcinoma (HCC) is a highly malignant tumor with an extremely poor prognosis. Our preliminary study indicated that bufalin could restrain the proliferation of human hepatoma BEL-7402 cells in a time- and dose-dependent manner. In the present study, the colony formation assay, the Transwell invasion assay, the western blot analysis and the immunofluorescence method were respectively used to investigate the effect and mechanism of bufalin against HCC cell invasion and metastasis. We found that: i) bufalin had significant inhibitory effect on the cell proliferation of BEL-7402 cells; ii) bufalin markedly inhibited the migration and invasion of BEL-7402 cells; iii) bufalin could suppress the phosphorylation of GSK-3β Ser9 site in BEL-7402 cells, decrease the expression of β-catenin, cyclin D1, metalloproteinases-7 (MMP-7) and cyclooxygenase-2 (COX-2) in the cytoplasm, and increase the expression of E-cadherin and β-catenin on the cell membrane; and iv) the expression of α-fetoprotein significantly decreased and the expression of albumin increased in BEL-7402 cells after bufalin was used. Our results indicate that: i) bufalin can regulate the expression of associated factors in Wnt/β-catenin signaling pathway of BEL-7402 cells through suppressing the phosphorylation of GSK-3β Ser9 site; ii) bufalin can strengthen intercellular E-cadherin/β-catenin complex to control epithelial-mesenchymal transition; and iii) bufalin can reverse the malignant phenotype and promote the differentiation and maturation by regulating the AFP and ALB expression in BEL-7402 cells. These are very important mechanisms of bufalin on the inhibition of the invasion and metastasis of HCC cells.
Integrin-linked kinase (ILK), a highly conserved intracellular protein of serine/threonine protein kinase activities, which is associated with the integrin and growth factor receptor signaling pathway, is involved in the regulation of cell proliferation, apoptosis, differentiation, migration and epithelial-mesenchymal transition (EMT). Findings of a previous study showed that ILK overexpression was strongly correlated with a more aggressive tumor phenotype, recurrence and poor survival for oral squamous cell carcinoma (OSCC) patients, as well as some EMT markers. In order to investigate the underlying mechanisms involved, a lentivirus-mediated short hairpin RNA (shRNA) was employed to downregulate ILK. The results showed that the knockdown of ILK inhibited cell growth, adhesion and invasion ability in vitro, and OSCC cells deficient of ILK were blocked in the S phase and underwent apoptosis. Additionally, ILK shRNA inhibited EMT by impairing the expression of Snail, Slug and Twist2 and enhacning E-cadherin expression. ILK shRNA suppressed the phosphorylation of downstream signaling targets Akt and GSk-3β. In addition, the knockdown of ILK inhibited tumor growth, invasion and metastasis of xenograft tumors in vivo. These results suggested that ILK is a promising therapeutic target for the treatment of OSCC.
Endotoxin can stimulate inflammatory cytokine release from monocytes/macrophages and result in septic shock. Glycyrrhetinic acid (GA), the main bioactive component of licorice, possesses substantial anti-inflammatory activity. Here, we explored effect of 11-deoxy-18α-glycyrrhetinic acid-30-ethyl ester (DGAEE), a newly synthesized derivative of GA, on septic shock. DGAEE and its main metabolite 11-deoxy-18α-glycyrrhetinic acid (DGA) significantly alleviated septic shock as evidenced by improvements of survival rates, lung histopathological changes and wet/dry ratio in lipopolysaccharide (LPS)/D-galactosamine-stimulated mice, and decreased blood pressure in LPS/D-galactosamine-stimulated rats. The two compounds decreased serum levels of NO, TNF-α, IL-6, IL-1β, and increased the level of IL-10 more potently in mice. In LPS-stimulated RAW 264.7 cells, DGA but not DGAEE showed marked regulation of NO, TNF-α, IL-6 and IL-10 levels, suggesting that DGAEE display anti-shock effect by DGA rather than itself. Moreover, the neutralizing antibody against IL-10 markedly prohibited the inhibitory effect of DGA on the production of cytokines from RAW 264.7 cells, and AS101 (an inhibitor of IL-10 biosynthesis) almost completely reversed the anti-shock effect of DGA in mice. In addition, DGA did not affect activation of NF-κB-p65 and p38 MAPK as well as IκBα degradation, but moderately reduced activation of ERK and JNK, and markedly increased phosphorylation of GSK3β in LPS-stimulated RAW 264.7 cells. LY294002 (an inhibitor of GSK3β phosphorylation) and LiCl (an inhibitor of GSK3β activity) diminished and potentiated increase of IL-10 levels by DGA, respectively. In conclusion, DGAEE alleviates septic shock through DGA in an IL-10-dependent manner, and the mechanism is related to inactivation of GSK3β.
MiRNAs play important roles in tumorigenesis. This study focused on exploring the effects and regulation mechanism of miRNA-137 on the biological behaviors of gastric cancer. Total RNA was extracted from tissues of 100 patients with gastric cancer and from four gastric cancer cell lines. Expression of miR-137 was detected by real-time PCR from 100 patients. The effects of miR-137 overexpression on gastric cancer cells' proliferation, apoptosis, migration and invasion ability were investigated in vitro and in vivo. The target gene of miR-137 was predicted by Targetscan on line software, screened by dual luciferase reporter gene assay and demonstrated by western blot. As a result, the expression of miR-137 was significant reduced in gastric cancer cell line HGC-27, HGC-803, SGC-7901 and MKN-45 as well as in gastric cancer tissues compared with GES-1 cell or matched adjacent non-neoplastic tissues (p<0.001). The re-introduction of miR-137 into gastric cancer cells was able to inhibit cell proliferation, migration and invasion. The in vivo experiments demonstrated that the miR-137 overexpression can reduce the gastric cancer cell proliferation and metastasis. Bioinformatic and western blot analysis indicated that the miR-137 acted as tumor suppressor roles on gastric cancer cells through targeting AKT2 and further affecting the Bad and GSK-3β. In conclusion, the miR-137 which is frequently down-regulated in gastric cancer is potentially involved in gastric cancer tumorigenesis and metastasis by regulating AKT2 related signal pathways.
It has been demonstrated that ATP-sensitive potassium (KATP) channel activation has neuroprotective effects against neuronal damage induced by hypoxia, ischemia or metabolism stress. This study investigated the multiply protective effects of KATP channel opener nicorandil against neurotoxicity in SH-SY5Y cells transiently transfected with Swedish mutant APP (APPsw) and also the potential involvement of PI3K/Akt/GSK-3β pathway. Cells were treated with nicorandil (1 mM) for 24 h with and without glibenclamide (10 μM), a KATP channel inhibitor. Then the cells were collected for Hoechst33342, biochemical assays, real-time PCR, western blot and ELISA assay. Our results showed that nicorandil reduced apoptosis and decreased oxidative stress. Moreover, nicorandil down regulated APP695 mRNA and APP695 protein expression, also reduced Aβ1-42 levels in the medium. In addition, nicorandil increased the protein levels of p-Akt and p-GSK-3β by PI3K activation. Applying a PI3K inhibitor, LY294002 blocked the protection. These findings suggest nicorandil to be a potential therapeutic agent to treat Alzheimer's disease (AD).
The embryonic stem cell (ESC)-enriched miR-294/302 family and the somatic cell-enriched let-7 family stabilizes the self-renewing and differentiated cell fates, respectively. The mechanisms underlying these processes remain unknown. Here we show that among many pathways regulated by miR-294/302, the combinatorial suppression of epithelial-mesenchymal transition (EMT) and apoptotic pathways is sufficient in maintaining the self-renewal of ESCs. The silencing of ESC self-renewal by let-7 was accompanied by the upregulation of several EMT regulators and the induction of apoptosis. The ectopic activation of either EMT or apoptotic program is sufficient in silencing ESC self-renewal. However, only combined but not separate suppression of the two programs inhibited the silencing of ESC self-renewal by let-7 and several other differentiation-inducing miRNAs. These findings demonstrate that combined repression of the EMT and apoptotic pathways by miR-294/302 imposes a synergistic barrier to the silencing of ESC self-renewal, supporting a model whereby miRNAs regulate complicated cellular processes through synergistic repression of multiple targets or pathways.
Inflammatory damage plays an important role in cerebral ischemic pathogenesis and may represent a target for treatment. Evodiamine (Evo) has been proved to elicit a variety of biological effects through its anti-inflammatory property in the treatment of infectious disease, Alzheimer's disease and hypoxia-induced inflammatory response. Whether this protective effect applies to cerebral ischemic injury, we therefore investigated the potential neuroprotective role of Evo and the underlying mechanisms. Male Institute of Cancer Research (ICR) mice were subjected to permanent middle cerebral artery occlusion (pMCAO) and randomly divided into five groups: Sham (sham-operated + 1% DMSO + 0.5% tween80), pMCAO (pMCAO + 0.9% saline), Vehicle (pMCAO + 1% DMSO + 0.5% tween80), Evo-L (Vehicle + Evo 50 mg/kg) and Evo-H (Vehicle + Evo 100 mg/kg) groups. Evo was administered intragastrically twice daily for 3 days, and once again 30 min before mouse brain ischemia was induced by pMCAO. Neurological deficit, brain water content and infarct size were measured at 24 h after stroke. The expression of pAkt, pGSK3β, NF-κB and claudin-5 in ischemic cerebral cortex was analyzed by western blot and qRT-PCR. Compared with Vehicle group, Evo significantly ameliorated neurological deficit, brain water content and infarct size, upregulated the expression of pAkt, pGSK3β and claudin-5, and downregulated the nuclear accumulation of NF-κB (P < 0.05). Evo protected the brain from ischemic damage caused by pMCAO; this effect may be through upregulation of pAkt, pGSK3β and claudin-5, and downregulation of NF-κB expression.
Tenuifoliside A (TFSA) is a bioactive oligosaccharide ester component of Polygala tenuifolia Wild, a traditional Chinese medicine which was used to manage mental disorders effectively. The neuroprotective and anti-apoptotic effects of TFSA have been demonstrated in our previous studies. The present work was designed to study the molecular mechanism of TFSA on promoting the viability of rat glioma cells C6. We exposed C6 cells to TFSA (or combined with ERK, PI3K and TrkB inhibitors) to examine the effects of TFSA on the cell viability and the expression and phosphorylation of key proteins in the ERK and PI3K signaling pathway. TFSA increased levels of phospho-ERK and phospho-Akt, enhanced release of BDNF, which were blocked by ERK and PI3K inhibitors, respectively (U0126 and LY294002). Moreover, the TFSA caused the enhanced phosphorylation of cyclic AMP response element binding protein (CREB) at Ser133 site, the effect was revoked by U0126, LY294002 and K252a. Furthermore, when C6 cells were pretreated with K252a, a TrkB antagonist, known to significantly inhibit the activity of brain-derived neurotrophic factor (BDNF), blocked the levels of phospho-ERK, phospho-Akt and phosphor-CREB. Taking these results together, we suggested the neuroprotection of TFSA might be mediated through BDNF/TrkB-ERK/PI3K-CREB signaling pathway in C6 glioma cells.
Proteasome inhibition can induce abnormal accumulation and phosphorylation of microtubule-associated protein tau. The major function of tau protein is to promote microtubules assembly and stabilization, and abnormal tau protein would disturb its microtubule-binding function. In this study, proteasome inhibitor MG132 was used to treat hippocampal slices to explore the role and mechanism of Akt/glycogen synthase kinase-3β (GSK-3β) in proteasome inhibition-induced tau abnormality. During the culture period, we measure the lactate dehydrogenase (LDH) content to assay the viability of hippocampal slices. Following 2.5 and 5 μM MG132 treatment for 6 h, we detected the expression, phosphorylation modification, and microtubule-binding function of tau protein of slices. We also analyzed the changed activities of glycogen synthase kinase-3β (GSK-3β) and protein kinase B (PKB/Akt) and the level of heat shock protein 90 (Hsp90) in the process. In addition, co-immunoprecipitation was used to investigate the interaction between Akt and Hsp90, Akt and protein phosphatase-2A (PP2A) in the MG132-treated organotypic hippocampal slices. Our results indicated that proteasome inhibition led to degradation obstacles and abnormal phosphorylation of tau protein. The downregulated Akt/GSK-3β signaling pathway might be responsible for the abnormal phosphorylation of tau protein at multiple sites which further reduced the microtubule-binding function of tau protein. Furthermore, proteasome inhibition decreased the binding capacity of Akt-Hsp90 while increased the Akt-PP2A binding ability which mediated Akt inactivity. This current study establishes a hippocampal slice model targeting Akt/GSK-3β signaling pathway to explore the pivotal role of proteasome inhibition in tau pathology.
Chronic noise exposure has been associated with abnormalities in glutamate (Glu)-NMDAR signaling and tau hyperphosphorylation. However, further studies are necessary to clarify potential causal relationships. The aim of the present study was to evaluate the role of NMDA receptors in noise-induced tau hyperphosphorylation in the rat hippocampus and prefrontal cortex. Male Wistar rats were randomly divided into three groups in the present study: control with isotonic saline instillation (n=10); noise exposure (100 dB SPL white noise, 4h/d × 14d) and treated with saline (n=10); and noise exposure and treated with MK-801 (0.5mg/kg, intraperitoneally; n=10). The levels of tau phosphorylated at Ser202 and Ser396, and proteins involved in hyperphosphorylation, namely glycogen synthase kinase 3β (GSK3β) and protein phosphatase 2A (PP2A), were measured in the hippocampus and prefrontal cortex (PFC) after the last noise exposure. We showed that phosphorylated tau levels were enhanced in noise-exposed-rat hippocampus and PFC. MK-801 decreased the hyperphosphorylation of tau at Ser202 and Ser396 sites in the hippocampus and PFC. Furthermore, MK-801 reversed noise-induced GSK3β overexpression but had no significant effect on PP2A levels. This suggests that MK-801 protects against chronic-noise-induced tau hyperphosphorylation in the hippocampus and PFC. These findings demonstrate that Glu-NMDAR signaling may be involved in triggering aberrant tau hyperphosphorylation in the hippocampus and PFC after chronic noise exposure.
Ketamine may produce rapid and sustained antidepressant effects. Despite the fact that the detailed underlying mechanism remains unknown, recent studies have suggested the involvement of the mammalian target of rapamycin (mTOR) pathway and glycogen synthase kinase-3 (GSK-3) signal, respectively, in the process of ketamine exerting antidepressant actions. This study aimed to investigate the mechanism by which ketamine phosphorylates GSK-3β in the rat prefrontal cortex (PFC) via applying vehicle or the antagonists of mTOR signalling pathway proteins including PI3K/Akt, mTOR and p70S6 kinase to the rats in the forced swimming test (FST) prior to ketamine administration, and subsequently observing the levels of phosphorylated GSK-3β, mTOR and p70S6K in rat PFC as well as the immobility time of rats in the FST. Our results revealed that compared to treatment with vehicle, ketamine increased the levels of phosphorylated GSK-3β in rat PFC (p < 0.05), which was attenuated by PI3K/Akt antagonist pretreatment (p < 0.05), but could not be affected by mTOR antagonist or p70S6K antagonist pretreatment. In addition, all the antagonists reversed the ketamine-induced increases in the phosphorylation of mTOR and p70S6K (p < 0.05). They also all abolished the rapid-acting antidepressant actions of ketamine demonstrated by the increased immobility time of rats in the FST. In conclusion, Akt mediates the phosphorylation of GSK-3β in rat PFC during the process of ketamine exerting rapid antidepressant actions.
Recent evidence indicates that severe abnormalities in brain glucose/energy metabolism and insulin signaling have been documented to take a pivotal role in early sporadic Alzheimer's disease pathology. It has been reported that naringenin (NAR), derived from citrus aurantium, exhibits antioxidant potential and protects the brain against neurodegeneration. The current study was designed to further investigate the protective effect of the NAR on neurodegeneration in a rat model of AD induced by an intracerebroventricular (ICV) injection of streptozotocin (STZ), and to determine whether this neuroprotective effect was associated with brain insulin signaling. Rats were injected bilaterally with ICV-STZ (3 mg/kg), while sham rats received the same volume of vehicle and then supplemented with NAR (25, 50 mg, 100 mg/kg, respectively) for 3 weeks. The ICV-STZ injected rats did not have elevated blood glucose levels. 21 days following ICV-STZ injection, rats treated with NAR had better learning and memory performance in the Morris water maze test compared with rats treated with saline. We demonstrated that NAR increased the mRNA expression of INS and INSR in cerebral cortex and hippocampus. In addition, NAR reversed ICV-STZ induced Tau hyper-phosphorylation in both hippocampus and cerebral cortex through downregulation of glycogen synthase kinase-3β (GSK-3β) activity, a key kinase in the insulin signaling. Brain levels of Abeta, which were elevated in ICV-STZ rats, were significantly reduced in NAR-treated rats via upregulation of insulin degrading enzyme. These effects were mediated by increased insulin and insulin receptors expression in the brain, suggesting that insulin sensitizer agents might have therapeutic efficacy in early AD.
Wogonin, a naturally occurring mono-flavonoid, has been reported to have tumor therapeutic potential and good selectivity both in vitro and in vivo. Herein, we investigated the anti-proliferation effects and associated mechanisms of wogonin in human colorectal cancer in vitro. The flow-cytometric analysis showed that wogonin induced a G1 phase cell cycle arrest in HCT116 cells in a concentration- and time-dependent manner. Meanwhile, the cell cycle-related proteins, such as cyclin A, E, D1, and CDK2, 4 were down-regulated in wogonin-induced G1 cell cycle arrest. Furthermore, we showed that the anti-proliferation and G1 arrest effect of wogonin on HCT116 cells was associated with deregulation of Wnt/β-catenin signaling pathway. Wogonin-treated cells showed decreased intracellular levels of Wnt proteins, and activated degradation complex to phosphorylated and targeted β-catenin for proteasomal degradation. Wogonin inhibited β-catenin-mediated transcription by interfering in the transcriptional activity of TCF/Lef, and repressing the kinase activity of CDK8 which has been considered as an oncogene involving in the development of colorectal cancers. Moreover, CDK8 siRNA-transfected HCT116 cells showed similar results to wogonin treated cells. Thus, our data suggested that wogonin induced anti-proliferation and G1 arrest via Wnt/β-catenin signaling pathway and it can be developed as a therapeutic agent against human colorectal cancer.
Malignant gliomas are the most common and most aggressive primary brain tumor, and for which differentiation therapy has emerged as a promising candidate strategy. In this study, we used in vitro and in vivo assays to examine the differentiation effects of wogonin, a major active constituent of Scutellaria baicalensis, on glioma C6 and U251 cells. We found that wogonin can suppress cell proliferation and induce G0/G1 arrest under a concentration-dependent manner. Wogonin also triggered significant reduction in the G1 cell-cycle regulatory proteins cyclin D1, cyclin-dependent kinase 2 and 4 along with overexpression of cell-cycle inhibitory proteins p27. Immunofluorescence and western blot analysis indicated that wogonin increased the expression of lineage-specific differentiation marker glial fibrillary acidic protein (GFAP). In mechanisms, we verified that wogonin significantly diminished the phosphorylated level of protein kinase B (AKT), and maintenance of low β-catenin expression level was dependent on glycogen synthase kinase 3β (GSK3β) activation at Ser9. Blocking GSK3β/β-catenin pathway was required for wogonin-induced proliferation inhibition and terminal differentiation by using canonical activator lithium chloride (LiCl) and inhibitor dickkopf-1 (Dkk1). Moreover, intravenous administration of wogonin delayed the growth of C6 glioma in the intracranial tumor model. These findings provide the evidence and mechanistic support for wogonin-based differentiation therapies for malignant glioblastoma. Furthermore, inhibition of GSK3β/β-catenin pathway may be a key and requisite factor in glioma differentiation.
Excessive βAR stimulation is an independent factor in inducing pathological cardiac hypertrophy. Here, we report miR-145 regulates both expression and localization of GATA6, thereby protecting the heart against cardiomyocyte hypertrophy induced by isoproterenol (ISO). The protective activity of miR-145 was associated with down-regulation of ANF, BNP and β-MHC expression, a decreased rate of protein synthesis, inhibited cardiomyocyte growth and the modulation of several signaling pathways including ERK1/2, JNK and Akt-GSK3β. The anti-hypertrophic effect was abrogated by exogenous over-expression of transcription factor GATA6 which was further identified as a direct target of miR-145. In addition, GSK3β antagonists, LiCl and TDZD8, restored the nuclear accumulation of GATA6, which was attenuated by miR-145 Finally, we observed a dynamic pattern of miR-145 expression in ISO-treated NRCMs and in the hearts of TAC mice. Together, our results identify miR-145 as an important regulator in cardiac hypertrophy.
We described a prospective application of ligand-based virtual screening program SHAFTS to discover novel inhibitors for p90 ribosomal S6 protein kinase 2 (RSK2). Taking the putative 3D conformations of two weakly binding RSK2 NTKD inhibitors as query templates, SHAFTS was used to perform 3D similarity based virtual screening because of a lack of crystal structure of RSK2 protein, thus leading to the identification of several novel scaffolds that would have been missed by conventional 2D fingerprint methods. The most potent hit compounds show low micromolar inhibitory activities against RSK2. In particular, one of the hit compounds exhibits potent antimigration activity against the MDA-MB-231 tumor cell. The results exemplified SHAFTS' application in active enrichment and scaffold hopping, which is of general interest for lead identification in drug discovery endeavors and also provides novel scaffolds that lay the foundation for uncovering new RSK2 regulatory mechanisms.
Astrocyte-elevated gene-1 (AEG-1) plays an important role in diverse cancers and its up-regulation is associated with poor survival of patients. However, the status of AEG-1 expression and its significance in gastric cancer are still unclear. In this study, the expression of AEG-1 was studied in different gastric cancer cell lines and gastric cancer tissues. Expression of AEG-1 was significantly higher in gastric cancer tissues than that in normal tissues. Overexpression of AEG-1 was found in 62.9% of gastric cancers and significantly associated with TNM stage and Ki-67 proliferation index (P < 0.01). For survival study, overexpression of AEG-1 was significantly associated with poor survival (P < 0.01). Further multivariate analysis suggested that AEG-1 overexpression was an independent prognostic factor for the disease. We demonstrated that inhibition of AEG-1 expression by specific siRNA clearly inhibited SGC-7901 cell growth and enhanced cell apoptosis (P < 0.01). Inhibition of AEG-1 reduced phosphorylation of AKT and glycogen synthase kinase (GSK)-3β (Ser 9) and decreased the level of β-catenin, lymphoid enhancer binding factor 1 (LEF1), and Cyclin D1. This indicated that AEG-1 may play a role in Wnt/β-catenin-mediated cancer progression. Taken together, overexpression of AEG-1 could be a useful prognostic factor in patients with gastric cancer. Targeted inhibition of AEG-1 may provide a novel therapeutic strategy for gastric cancer.
Bone marrow-derived mesenchymal stem cells (MSCs) have been demonstrated to be able to differentiate into epithelial lineage, but the precise mechanisms controlling this process are unclear. Our aim is to explore the roles of Wnt/beta-catenin in the epithelial differentiation of MSCs. Using indirect co-culture of rat MSCs with rat airway epithelial cells (RTE), MSCs expressed several airway epithelial markers (cytokeratin 18, tight junction protein occudin, cystic fibrosis transmembrance regulator). The protein levels of some important members in Wnt/beta-catenin signaling were determined, suggested down-regulation of Wnt/beta-catenin with epithelial differentiation of MSCs. Furthermore, Wnt3alpha can inhibit the epithelial differentiation of MSCs. A loss of beta-catenin induced by Dickkopf-1 can enhance MSCs differentiation into epithelial cells. Lithium chloride transiently activated beta-catenin expression and subsequently decreased beta-catenin level and at last inhibited MSCs to differentiate into airway epithelium. Taken together, our study indicated that RTE cells can trigger epithelial differentiation of MSCs. Blocking Wnt/beta-catenin signaling may promote MSCs to differentiate towards airway epithelial cells.
AIM:
Tuberous sclerosis complex 2 (TSC2), a tumor suppressor, may play an essential role in the regulation of cell growth and cell survival under energy stress conditions. In addition, TSC2 may act in concert with Wnt and energy signals by additional phosphorylation of glycogen synthase kinase 3β (GSK3β) to regulate cell growth. The expression levels and function of TSC2 and GSK3β in hepatocellular carcinoma (HCC) remain unclear.
METHODS:
The protein levels of TSC2 and GSK3β were measured by immunohistochemistry in normal liver (n = 20), HCC (n = 80) and pericancerous tissues (n = 80). The correlations between TSC2, and GSK3β levels, clinicopathological features and patient survival were also analyzed.
RESULTS:
The protein levels of TSC2 and GSK3β in HCC tissues were significantly lower than that in normal liver tissues and pericancerous tissues (P < 0.05). Decreased TSC2 and GSK3β expression was found to be significantly correlated with advanced clinicopathological characteristics and poor prognosis. The results also showed that TSC2 protein levels were associated with GSK3β expression in HCC specimens.
CONCLUSION:
This is the first demonstration that the decreases in TSC2 and GSK3β levels may be associated with vascular invasion, histological grade and tumor-node-metastasis classification.
© 2013 The Japan Society of Hepatology.
RATIONALE:
Xanthoceraside, a novel triterpenoid saponin extracted from the fruit husks of Xanthoceras sorbifolia Bunge, reverses cognitive deficits in intracerebroventricular injection of Aβ25-35 or Aβ1-42 mice. However, whether xanthoceraside has a positive effect on hyperphosphorylated tau protein remains unclear.
OBJECTIVES:
We investigated the effects of xanthoceraside on behavioural impairments induced by intracerebroventricular injection of streptozotocin (STZ) in rats and its potential mechanisms.
MATERIALS AND METHODS:
The rats were administered with xanthoceraside (0.06, 0.12 or 0.24 mg/kg) or vehicle once daily after STZ intracerebroventricular injections. The Y-maze test and novel object recognition test were performed 21 and 22 days after the second STZ injection, respectively. The levels of hyperphosphorylated tau, phosphatidylinositol-3-kinase (PI3K)/serine/threonine protein kinase B (Akt), glycogen synthase kinase-3β (GSK-3β), protein phosphatase 1 (PP-1) and protein phosphatase 2A (PP-2A) were also tested by Western blot.
RESULTS:
Xanthoceraside treatment significantly attenuated learning and memory impairments and reduced the level of STZ-induced hyperphosphorylated tau protein. Xanthoceraside also enhanced PP-2A and PP-1 expressions, increased PI3K (p85) and Akt (Ser473) phosphorylation and decreased GSK-3β (tyr216) phosphorylation.
CONCLUSIONS:
Xanthoceraside has protective effect against learning and memory impairments and inhibits tau hyperphosphorylation in the hippocampus, possibly through the inhibition of the PI3K/Akt-dependent GSK-3β signalling pathway and an enhancement of phosphatases activity.
BACKGROUND:
Acquired resistance to epidermal growth factor receptor tyrosine kinase inhibitors (EGFR-TKIs) presents great challenges in the treatment of non-small cell lung cancer (NSCLC) patients, while the mechanisms are still not well understood. The β-catenin signaling pathway has been found to be associated with chemoresistance and can activate the EGFR and its downstream pathways. This study aimed to investigate the role of β-catenin in acquired resistance to EGFR-TKIs in NSCLC cell lines.
METHODS:
The expression and transcriptional activity of β-catenin were measured in both the NSCLC cell line PC9 and its sub-line PC9/AB(2) which has acquired resistance to gefitinib. Knockdown and overexpression of β-catenin in the PC9/AB(2) and PC9 cells were performed. The cell survival rate and the activation of the EGFR and its downstream pathways were detected in the two cell lines after transfection.
RESULTS:
Nuclear translocation of β-catenin was increased in the PC9/AB(2) cells and the baseline expression of members of the β-catenin signaling pathway was also higher in the PC9/AB(2) cells. Knocking down the expression of β-catenin increased the sensitivity of the PC9/AB(2) cells to gefitinib by blocking the activation of the EGFR downstream pathways, while β-catenin overexpression improved PC9 cells resistance to gefitinib by enhancing the activation of the EGFR and its downstream signaling.
CONCLUSION:
β-catenin plays an important role in acquired resistance to EGFR-TKIs in NSCLC cell lines and may be a potential therapeutic target for NSCLC patients who have failed to respond to targeted therapy.
Crown Copyright © 2013. Published by Elsevier Ltd. All rights reserved.
PURPOSE:
This study was to investigate the clinicopathologic significance and potential role of HOXB7 in the development and progression of colorectal cancer (CRC).
EXPERIMENTAL DESIGN:
The relationship between HOXB7 expression and clinical characteristics of CRC was analyzed in 224 paraffin-embedded archived CRC specimens by immunohistochemistry (IHC). The effects of HOXB7 on cell growth and proliferation, as well as on tumorigenesis, were examined both in vitro and in vivo, using MTT assay, colony formation assay, cell cycle analysis, soft agar assay, and tumorigenesis in nude mice. Western blotting and real-time reverse transcriptase-PCR were performed to examine the impact of HOXB7 on the PI3K/Akt and MAPK signaling pathways.
RESULTS:
HOXB7 protein level was significantly correlated with advanced Dukes stage (P < 0.001), T stage (P = 0.012), distant metastasis (P = 0.042), higher proliferation index (P = 0.007) and poor survival of patients (P = 0.005). Enforced expression of HOXB7 in CRC cell lines significantly enhanced cell growth, proliferation and tumorigenesis. Conversely, knockdown of HOXB7 caused an inhibition of cell growth, proliferation, and tumorigenesis. We also showed that HOXB7 accelerated G(0)-G(1) to S-phase transition concomitantly with upregulation of cyclin D1 and downregulation of p27Kip1. On the contrary, knockdown of HOXB7 caused G(1)-S-phase arrest, downregulation of cyclin D1 and upregulation of p27Kip1. Enforced expression of HOXB7 could enhance PI3K/AKT and MAPK pathway activity.
CONCLUSION:
Our findings suggest that HOXB7 protein, as a valuable marker of CRC prognosis, plays an important role in the development and progression of human CRC.
©2011 AACR.