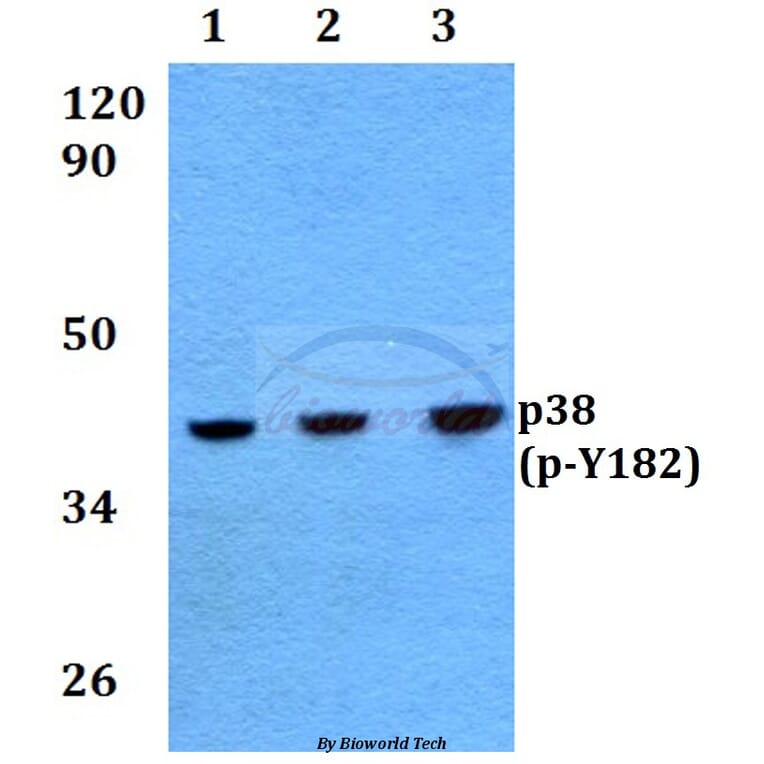
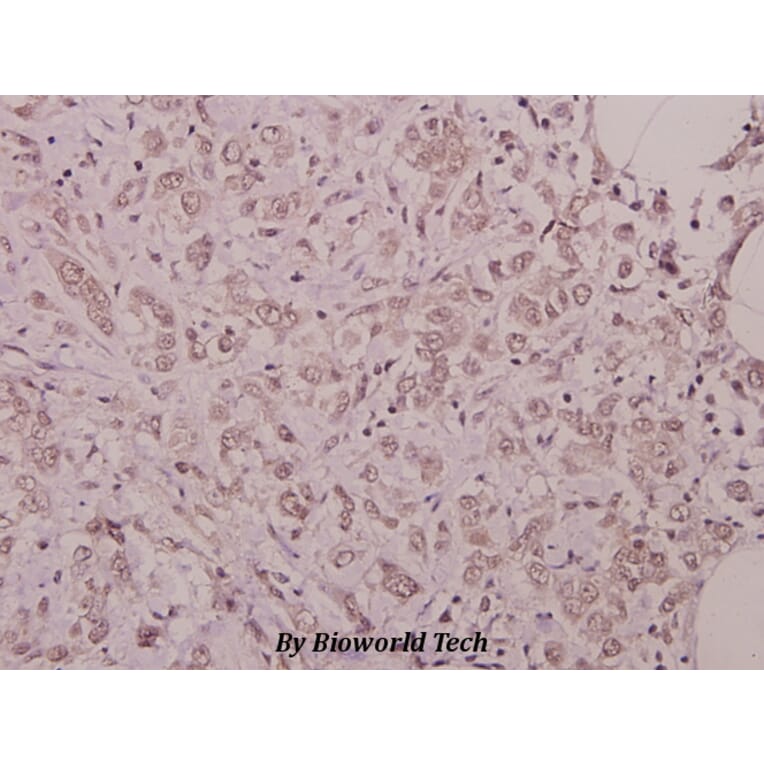
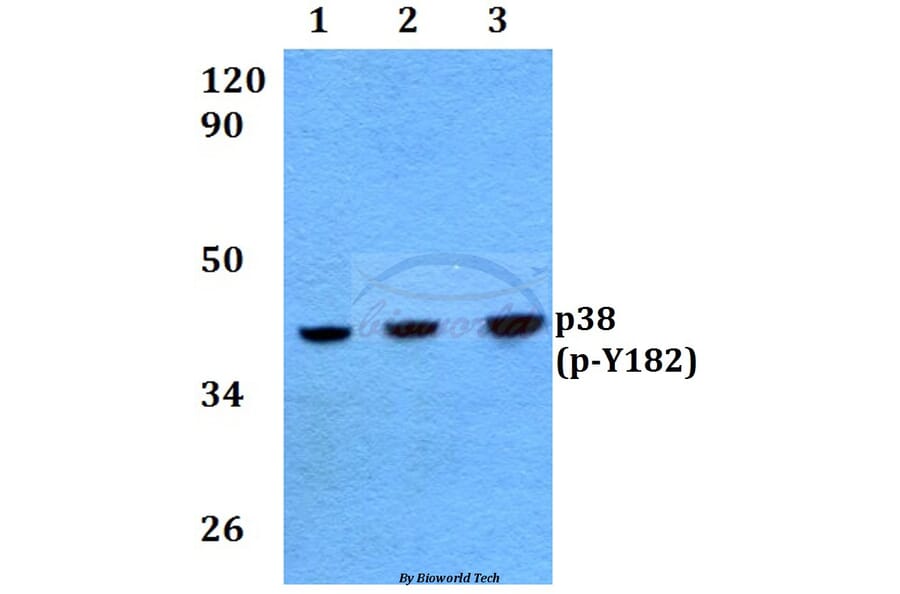
Unconjugated
Phloretin (Ph) existing in apples, pears and various vegetables is known to have antitumor activities in several cancer cell lines. However, little is known about its effect on human lung cancer cells. The aim of the present study was to see whether Ph could induce apoptosis of non-small cell lung cancer (NSCLC) cells, and explore the possible underlying mechanism of action. We found that Ph markedly induced cell apoptosis of NSCLC cell line A549, and inhibited the migration of A549 cells in a dose-dependent manner. The expression level of BAX, cleaved caspase-3 and -9, and degraded form of PARP was increased and Bcl-2 was decreased after Ph treatment. In addition, the phosphorylation of P38 MAPK, ERK1/2 and JNK1/2 was increased in a dose‑dependent manner in parallel with Ph treatment. Inhibition of P38 MAPK and JNK1/2 by specific inhibitors significantly abolished the Ph-induced activation of the caspase-3 and -9. In vivo tumor-suppression assay further indicated that Ph (20 mg/kg) displayed a more significant inhibitory effect on A549 xenografts in tumor growth. All these findings indicate that Ph is able to inhibit NSCLC A549 cell growth by inducing apoptosis through P38 MAPK and JNK1/2 pathways, and therefore may prove to be an adjuvant to the treatment of NSCLC.
Cathepsin B (CB), an important proteinase that participates in joint destruction in rheumatoid arthritis (RA), exhibits higher expression in fibroblast-like synoviocyte (FLS) of abnormal proliferative synovial tissues. Whether and how it affects the biological behaviours of RA-FLS, such as migration and invasion, are poorly understood. In the present study, CB expression in synovial tissues of patients with RA and ostearthritis (OA) were measured by quantitative polymerase chain reaction (qPCR) and immunohistochemistry (IHC), respectively. Stable depletion of endogenous CB was achieved by small interfering RNA (siRNA) transfection, and decrease of CB activity was acquired by using its specific inhibitor (CA074Me). The effects of CA074Me and RNA interference (RNAi) treatments on proliferation, migration, invasion, matrix metalloproteinase (MMP)-2/-9 expression, focal adhesion kinase (FAK) activation, and mitogen-activated protein kinases (MAPKs) phosphorylation of FLS were analysed. In RA synovial tissues, CB was expressed at elevated levels compared with OA synovial tissues. CA074Me could inhibit invasion of FLS obtained from RA patients in an ex-vivo invasion model. CA074Me and siRNA treatments suppressed the migration and invasion of FLS, reduced the activity, expression and mRNA level of MMP-2, restrained the activation of FAK and reduced the expression of F-actin. Moreover, CA074Me decreased the phosphorylation of P38 MAPK and c-Jun N-terminal kinase (JNK) in FLS, while siCB treatment reduced the phosphorylation of P38 but not JNK. CB substantially contributes to the invasive phenotype of FLS that leads to joint destruction in RA. This proteinase may show promise as a therapeutic target in inflammatory arthritis.
It has been documented in in vitro studies that zinc oxide nanoparticles (ZnO NPs) are capable of inducing oxidative stress, which plays a crucial role in ZnO NP-mediated apoptosis. However, the underlying molecular mechanism of apoptosis in neurocytes induced by ZnO NP exposure was not fully elucidated. In this study, we investigated the potential mechanisms of apoptosis provoked by ZnO NPs in cultured primary astrocytes by exploring the molecular signaling pathways triggered after ZnO NP exposure. ZnO NP exposure was found to reduce cell viability in MTT assays, increase lactate dehydrogenase (LDH) release, stimulate intracellular reactive oxygen species (ROS) generation, and elicit caspase-3 activation in a dose- and time-dependent manner. Apoptosis occurred after ZnO NP exposure as evidenced by nuclear condensation and poly(ADP-ribose) polymerase-1 (PARP) cleavage. A decrease in mitochondrial membrane potential (MMP) with a concomitant increase in the expression of Bax/Bcl-2 ratio suggested that the mitochondria also mediated the pathway involved in ZnO NP-induced apoptosis. In addition, exposure of the cultured cells to ZnO NPs led to phosphorylation of c-Jun N-terminal kinase (JNK), extracellular signal-related kinase (ERK), and p38 mitogen-activated protein kinase (p38 MAPK). Moreover, JNK inhibitor (SP600125) significantly reduced ZnO NP-induced cleaved PARP and cleaved caspase-3 expression, but not ERK inhibitor (U0126) or p38 MAPK inhibitor (SB203580), indicating that JNK signaling pathway is involved in ZnO NP-induced apoptosis in primary astrocytes.
Tumor necrosis factor-related apoptosis-inducing ligand (TRAIL) is a promising candidate for the treatment of cancer, because it preferentially induces apoptosis in numerous cancer cells with little or no effect on normal cells. 5,7-Dihydroxyflavone is a dietary flavonoid commonly found in many plants. Here we show that the combined treatment with 5,7-dihydroxyflavone and TRAIL at subtoxic concentrations induced strong apoptotic response in human hepatocarcinoma HepG2 cells, acute leukemia Jurkat T cells, and cervical carcinoma HeLa cells. We further investigated the mechanisms by which 5,7-dihydroxyflavone augments TRAIL-induced apoptosis in HepG2 cells. 5,7-Dihydroxyflavone up-regulated the expression of pro-apoptotic protein Bax, attenuated the expression of anti-apoptotic proteins Bcl-2, Mcl-1, and IAPs, and reduced the phosphorylation levels of Akt and STAT3, weakening the anti-apoptotic signals thus facilitating the process of apoptosis. Moreover, 5,7-dihydroxyflavone and TRAIL were well tolerated in mice, and the combination of 5,7-dihydroxyflavone and TRAIL reduced tumor burden in vivo in a HepG2 tumor xenograft model. Interestingly, 5,7-dihydroxyflavone-mediated sensitization to TRAIL-induced cell death was not observed in normal human hepatocytes L-O2. These results suggest that the 5,7-dihydroxyflavone in combination with TRAIL might be used for cancer prevention and/or therapy.
The development of cardiac hypertrophy in response to increased hemodynamic load and neurohormonal stress is initially a compensatory response that may eventually lead to ventricular dilation and heart failure. Regulator of G protein signaling 5 (Rgs5) is a negative regulator of G protein-mediated signaling by inactivating Galphaq and Galphai, which mediate actions of most known vasoconstrictors. Previous studies have demonstrated that Rgs5 expresses among various cell types within mature heart and showed high levels of Rgs5 mRNA in monkey and human heart tissue by Northern blot analysis. However, the critical role of Rgs5 on cardiac remodeling remains unclear. To specifically determine the role of Rgs5 in pathological cardiac remodeling, we used transgenic mice with cardiac-specific overexpression of human Rgs5 gene and Rgs5-/- mice. Our results demonstrated that the transgenic mice were resistant to cardiac hypertrophy and fibrosis through inhibition of MEK-ERK1/2 signaling, whereas the Rgs5-/- mice displayed the opposite phenotype in response to pressure overload. These studies indicate that Rgs5 protein is a crucial component of the signaling pathway involved in cardiac remodeling and heart failure.
AIMS:
To investigate the effects and underlying mechanism of dexmedetomidine on the cultured human dendritic cells (DCs).
METHODS:
Human DCs and cytotoxic T lymphocytes (CTLs) were obtained from human cord blood mononuclear cells by density gradient centrifugation. Cultured DCs were divided into three groups: dexmedetomidine group, dexmedetomidine plus yohimbine (dexmedetomidine inhibitor) group and control group. DCs in the three groups were treated with dexmedetomidine, dexmedetomidine plus yohimbine and culture medium, respectively. After washing, the DCs were co-incubated with cultured CTLs. The maturation degree of DCs was evaluated by detecting (1) the ratios of HLA-DR-, CD86-, and CD80-positive cells (flow cytometry), and (2) expression of IL-12 and IL-23 (PCR and Elisa). The function of DCs was evaluated by detecting the proliferation (MTS assay) and cytotoxicity activity (the Elisa of IFN-γ) of CTLs. In addition, in order to explore the mechanisms of dexmedetomidine modulating DCs, α2-adrenergic receptor and its downstream signals in DCs were also detected.
RESULTS:
The ratios of HLA-DR-, CD86-, and CD80-positive cells to total cells were similar among the three groups (P>0.05). Compared to the control group, the protein levels of IL-12 and IL-23 in the culture medium and the mRNA levels of IL-12 p35, IL-12 p40 and IL-23 p19 in the DCs all decreased in dexmedetomidine group (P<0.05). In addition, the proliferation of CTLs and the secretion of IFN-γ also decreased in the dexmedetomidine group, compared with the control group (P<0.05). Moreover, these changes induced by dexmedetomidine in the dexmedetomidine group were reversed by α2-adrenergic receptor inhibitor yohimbine in the dexmedetomidine plus yohimbine group. It was also found the decrease of mRNA levels of IL-12 p35, IL-12 p40 and IL-23 p19 in the dexmedetomidine group could be reversed by ERK1/2 or AKT inhibitors.
CONCLUSION:
Dexmedetomidine could negatively modulate human immunity by inhibiting the maturation of DCs and then decreasing the proliferation and cytotoxicity activity of CTLs. The α2-adrenergic receptors and its downstream molecules ERK1/2 and AKT are closely involved in the modulation of dexmedetomidine on DCs.
BACKGROUND:
The dipeptidyl peptidase-4 inhibitor sitagliptin, a new anti-diabetic medicine, is effective in treating type 2 diabetes mellitus by increasing the activation and duration of action of glucagon-like peptide-1. Since atherosclerosis is the main pathological feature of diabetic cardiovascular complications, it is important to investigate the anti-atherosclerotic effect of sitagliptin and explore the relevant mechanisms.
METHODS:
Male apolipoprotein-E-knockout mice were randomly divided into two groups and fed either high-fat diet (HFD) or HFD plus sitagliptin at a concentration of 0.3% for 16 weeks. Body weight, food intake, blood glucose, serum lipids and adhesion molecules were measured. The atherosclerotic plaque area and its histological composition were analyzed using Sudan staining and immunohistochemistry. The expression of inflammatory cytokines (monocyte chemoattractant protein (MCP)-1 and interleukin (IL)-6) and the activation of AMP-activated protein kinase (AMPK) and mitogen-activated protein kinase (MAPK) in the aortas were determined using quantitative polymerase chain reaction and western blot, respectively.
RESULTS:
Mice treated with sitagliptin developed fewer atherosclerotic plaques than the control group (7.64 ± 1.98% vs 12.91 ± 1.15%, p < 0.001), particularly in the aortic arch and abdominal aorta, where plaques were decreased 1.92- and 2.74-fold, respectively (p < 0.05 and p < 0.01). Sitagliptin significantly reduced the content of collagen fiber in plaques 1.2-fold (p < 0.05). Moreover, sitagliptin significantly reduced the expression of monocyte chemoattractant protein-1 and interleukin-6 in the aorta (p < 0.01 and p < 0.05), as well as the serum levels of soluble vascular cell adhesion molecule-1 and P-selectin (both p < 0.05). In addition, Sitagliptin induced phosphorylation of AMPK and Akt (p < 0.05 and p < 0.01), while suppressed phosphorylation of p38 and extracellular signal-regulated kinase (Erk) 1/2 (p < 0.05 and p < 0.01) in aortas.
CONCLUSIONS:
Our present study indicates that sitagliptin can reduce the area of the atherosclerotic lesion, possibly by regulating the AMPK and MAPK pathways and then reducing leukocyte -endothelial cell interaction and inflammation reactions. These actions are independent of weight loss and glucose-reducing effects.
The anti-cancer effects of dioscin have been widely reported. However, its effect on laryngeal cancer remains unknown. In the present paper, our results showed that dioscin markedly caused cell apoptosis and DNA damage, increased reactive oxygen species (ROS) level, induced S-phase arrest, and inhibited invasion of human laryngeal cancer HEp-2 and TU212 cells. Mechanism investigation showed that dioscin markedly up-regulated p53 level, and down-regulated cyclin-dependent kinase 2 (CDK2) and Cyclin A levels. In addition, dioscin significantly down-regulated the levels of p-ERK, Bcl-2, up-regulated the levels of p-JNK, p-p38, Bax, cleaved caspase-3/-9, and caused Cytochrome c release. Furthermore, U0126, an ERK1/2 inhibitor, markedly down-regulated Bcl-2 level, up-regulated the levels of Bax, cleaved caspase-3/9, and enhanced Cytochrome c release inducted by dioscin. While, SP600125 (one JNK inhibitor) and SB203580 (one p38 inhibitor) markedly up-regulated Bcl-2 level, down-regulated the levels of Bax, cleaved caspase-3/9, and obviously boosted Cytochrome c release induced by dioscin. Interestingly, dioscin also markedly down-regulated the levels of MMP2 and MMP9 associated with tumor invasion. Taken together, our study indicated that dioscin suppressed laryngeal cancer cells growth via inducting cell-cycle arrest, MAPK-mediated mitochondrial- derived apoptosis and inhibiting tumor invasion, which could be used as one potential candidate for the treatment of laryngeal cancer in the future.
Endotoxin can stimulate inflammatory cytokine release from monocytes/macrophages and result in septic shock. Glycyrrhetinic acid (GA), the main bioactive component of licorice, possesses substantial anti-inflammatory activity. Here, we explored effect of 11-deoxy-18α-glycyrrhetinic acid-30-ethyl ester (DGAEE), a newly synthesized derivative of GA, on septic shock. DGAEE and its main metabolite 11-deoxy-18α-glycyrrhetinic acid (DGA) significantly alleviated septic shock as evidenced by improvements of survival rates, lung histopathological changes and wet/dry ratio in lipopolysaccharide (LPS)/D-galactosamine-stimulated mice, and decreased blood pressure in LPS/D-galactosamine-stimulated rats. The two compounds decreased serum levels of NO, TNF-α, IL-6, IL-1β, and increased the level of IL-10 more potently in mice. In LPS-stimulated RAW 264.7 cells, DGA but not DGAEE showed marked regulation of NO, TNF-α, IL-6 and IL-10 levels, suggesting that DGAEE display anti-shock effect by DGA rather than itself. Moreover, the neutralizing antibody against IL-10 markedly prohibited the inhibitory effect of DGA on the production of cytokines from RAW 264.7 cells, and AS101 (an inhibitor of IL-10 biosynthesis) almost completely reversed the anti-shock effect of DGA in mice. In addition, DGA did not affect activation of NF-κB-p65 and p38 MAPK as well as IκBα degradation, but moderately reduced activation of ERK and JNK, and markedly increased phosphorylation of GSK3β in LPS-stimulated RAW 264.7 cells. LY294002 (an inhibitor of GSK3β phosphorylation) and LiCl (an inhibitor of GSK3β activity) diminished and potentiated increase of IL-10 levels by DGA, respectively. In conclusion, DGAEE alleviates septic shock through DGA in an IL-10-dependent manner, and the mechanism is related to inactivation of GSK3β.
Norisoboldine (NOR), the main active constituent of Radix Linderae, was previously demonstrated to ameliorate collagen-induced arthritis in rats through regulating the imbalance of T cells in intestines, which implied its therapeutic potential in inflammatory bowel disease. Here, we investigated the effect of NOR on ulcerative colitis (UC) induced by dextran sulfate sodium (DSS) in mice. Results showed that NOR (20, 40mg/kg) markedly reduced the symptoms of colitis, the levels of IL-1β and TNF-α, and the activation of ERK, p38 MAPK and NF-κB-p65. NOR only slightly decreased the levels of IFN-γ and IL-17A in mouse colons, but it dramatically increased the level of IL-10 at both protein and mRNA grades. Consistently, NOR increased the number of CD4(+)CD25(+)Foxp3(+) Treg cells more obviously than it decreased that of CD4(+)IL-17(+) Th17 cells in mesenteric lymph nodes (MLNs) and colonic lamina proprias (LPs) of colitis mice, and promoted the expression of Foxp3 mRNA in colon tissues. It could facilitate the in vitro differentiation of Treg cells from naive T cells and promote the phosphorylations of Smad2/3 in colon tissues of colitis mice. On the other hand, NOR did not affect the expressions of homing receptors CCR9 and α4β7 in SPs, and homing ligands CCL25 and Madcam-1 in MLNs and colonic LPs, suggesting that the increase of Treg cells in colons by NOR was not due to gut homing. In conclusion, NOR can ameliorate DSS-induced UC in mice, and the mechanisms involve reduction of pro-inflammatory cytokines and selective induction of Treg cells in colons.
The aim of this study was to explore the intracellular mechanisms underlying the cardiovascular toxicity of air particulate matter (PM) with an aerodynamic diameter of less than 2.5 µm (PM2.5) in a human umbilical vein cell line, EA.hy926. We found that PM2.5 exposure triggered reactive oxygen species (ROS) generation, resulting in a significant decrease in cell viability. Data from Western blots showed that PM2.5 induced phosphorylation of Jun N-terminal kinase (JNK), extracellular signal regulatory kinase (ERK), p38 mitogen-activated protein kinase (MAPK) and protein kinase B (AKT), and activation of nuclear factor kappa B (NF-κB). We further observed a significant increase in expressions of intercellular adhesion molecule-1 (ICAM-1) and vascular adhesion molecule-1 (VCAM-1) in a time- and dose-dependent manner. Moreover, the adhesion of monocytic THP-1 cells to EA.hy926 cells was greatly enhanced in the presence of PM2.5 . However, N-acetylcysteine (NAC), a scavenger of ROS, prevented the increase of ROS generation, attenuated the phosphorylation of the above kinases, and decreased the NF-κB activation as well as the expression of ICAM-1 and VCAM-1. Furthermore, ERK inhibitor (U0126), AKT inhibitor (LY294002) and NF-κB inhibitor (BAY11-7082) significantly down-regulated PM2.5 -induced ICAM-1 and VCAM-1 expression as well as adhesion of THP-1 cells, but not JNK inhibitor (SP600125) and p38 MAPK inhibitor (SB203580), indicating that ERK/AKT/NF-κB is involved in the signaling pathway that leads to PM2.5 -induced ICAM-1 and VCAM-1 expression. These findings suggest PM2.5 -induced ROS may function as signaling molecules triggering ICAM-1 and VCAM-1 expressions through activating the ERK/AKT/NF-κB-dependent pathway, and further promoting monocyte adhesion to endothelial cells.
The study aims to investigate the effects of protocatechuic acid (PCA) separated from Chinese herbs, on acute lung injury (ALI) induced by lipopolysaccharide (LPS) in mice. The mouse model was induced by intraperitoneal injection of LPS at the dose of 5mg/kg body weight. Three doses of PCA (30, 15, 5 mg/kg) were administered to mice with intraperitoneal injection one hour prior to LPS exposure. Six hours later after LPS administration, the effect of PCA on ALI mice was assessed via histopathological examination by HE staining, inflammatory cytokine production by ELISA assay and RT-PCR, p38MAPK and NF-κB activation by Western blot analysis. We found that PCA administration significantly ameliorated lung histopathological changes and decreased protein concentration in the bronchoalveolar lavage fluid. Furthermore, the overproduction of tumor necrosis factor-α (TNF-α) and interleukin-1β (IL-1β) was reduced by PCA. Additionally, PCA at the dose of 30 mg/kg could block the activation of p38MAPK and NF-κB signal pathways induced by LPS. In conclusion, our findings demonstrate that PCA possesses a protective effect on LPS-induced ALI in mice via suppression of p38MAPK and NF-κB signal pathways. Therefore, PCA may be useful in the therapy of lung inflammatory diseases, especially for ALI.
Urocortin (UCN) has exhibited antiinflammatory and neuroprotective effects on intracerebral hemorrhage (ICH). However, the underlying mechanisms are still not clear. Therefore, this study was aimed to investigate effects of UCN1 on ICH in vitro and in vivo and further explore the possible mechanism. ICH was induced by an infusion of autologous blood into the unilateral striatum of anesthetized male Sprague-Dawley rats. The rats were randomly divided into three groups (8 rats per group): sham ICH control group, ICH saline group and ICH UCN1 group. UCN1 was infused into the lateral ventricle after 1h post-ICH. Neurological deficits were evaluated by modified neurological severity score (mNSS). Brain edema was assessed using the dry/wet method. The neurological cell metabolic activity of N2a and SH-SY5Y was detected by CCK-8. The level of VEGF, JNK and p38 were determined by enzyme-linked immunosorbent assay and western blot. Post-treatment with UCN1 could improve neurological deficits and reduce brain edema. Moreover, UCN1 could increase the metabolic activity of neuron cells dose-dependently and these effects could be abolished by corticotropin-releasing factor receptor 2 (CRFR2) antagonist anti-Svg-30. Furthermore, the level of VEGF, JNK and p38 were up-regulated by post-treatment with UCN1 via CRFR2. The protective effects of UCN1 against ICH are possibly mediated by activating the phosphorylation of JNK and p38 and further increasing the level of VEGF via CRFR2.
The aim of the present paper was to investigate the effects and possible mechanisms of the total saponins from Dioscorea nipponica Makino (TSDN) against type 2 diabetes mellitus. Streptozotocin (STZ) with high-fat diet induced type 2 diabetes mellitus (T2DM) rats were treated with TSDN. Some biochemical parameters, target proteins and genes were investigated. The results showed that TSDN decreased the levels of food/water intake, fasting blood glucose and serum lipid parameters, ameliorated oral glucose and insulin tolerance test levels, markedly increased body weight and serum insulin, reduced excess free radicals and affected ossification and renal protection. Histopathological examination indicated that TSDN increased liver glycogen, decreased the production of lipid vacuoles and lightened liver damage. Further investigation showed that TSDN down-regulated the protein expressions of NF-κB, GRP78, ATF6, eIF2 and the levels of MAPK phosphorylation and up-regulated the protein expressions of IRS-1, GLUT-4, p-Akt and p-AMPK. In addition, TSDN obviously decreased the gene expressions of TNF-a, IL-6, PEPCK, G6Pase, GSK-3β and GSK-3β activity, and increased the gene expressions of PFK, PK and GK activity. These findings show the anti-diabetic activity of total saponins from D. nipponica Makino, which should be developed as a new potent drug for treatment of diabetes mellitus in future.
The crude powder of the fruit of Arctium lappa L. (ALF) has previously been reported to attenuate experimental colitis in mice. But, its main effective ingredient and underlying mechanisms remain to be identified. In this study, ALF was extracted with ethanol, and then successively fractionated into petroleum ether, ethyl acetate, n-butanol and water fraction. Experimental colitis was induced by dextran sulfate sodium (DSS) in mice. Among the four fractions of ALF, the ethyl acetate fraction showed the most significant inhibition of DSS-induced colitis in mice. The comparative studies of arctigenin and arctiin (the two main ingredients of ethyl acetate fraction) indicated that arctigenin rather than arctiin could reduce the loss of body weight, disease activity index and histological damage in the colon. Arctigenin markedly recovered the loss of intestinal epithelial cells (E-cadherin-positive cells) and decreased the infiltration of neutrophils (MPO-positive cells) and macrophages (CD68-positive cells). Arctigenin could down-regulate the expressions of TNF-α, IL-6, MIP-2, MCP-1, MAdCAM-1, ICAM-1 and VCAM-1 at both protein and mRNA levels in colonic tissues. Also, it markedly decreased the MDA level, but increased SOD activity and the GSH level. Of note, the efficacy of arctigenin was comparable or even superior to that of the positive control mesalazine. Moreover, it significantly suppressed the phosphorylation of MAPKs and the activation of NF-κB, including phosphorylation of IκBα and p65, p65 translocation and DNA binding activity. In conclusion, arctigenin but not arctiin is the main active ingredient of ALF for attenuating colitis via down-regulating the activation of MAPK and NF-κB pathways.
Oxidative stress-induced neuronal death has an important role in the pathogenesis of neurodegenerative disorders. The effects and mechanisms of action of the total flavonoids (TFs) from Rosa laevigata Michx fruit against hydrogen peroxide (H2O2)-induced oxidative injury in PC12 cells were investigated in this study. The results demonstrated that the TFs protected against cell apoptosis, DNA and mitochondrial damage caused by H2O2 based on single cell gel electrophoresis, in situ terminal deoxynucleotidyltransferase dUTP nick end labeling (TUNEL), flow cytometry and transmission electron microscope (TEM) assays. In addition, the TFs notably decreased cytochrome C release from mitochondria into the cytosol and intracellular Ca2+ levels, and diminished intracellular generation of reactive oxygen species (ROS). Furthermore, the TFs inhibited the phosphorylation levels of JNK, ERK and p38 MAPK as well as down-regulated the expressions of IL-1, IL-6, TNF-α, Fas, FasL, CYP2E1, Bak, caspase-3, caspase-9, p53, COX-2, NF-κB, AP-1, and up-regulated the expressions of Bcl-2 and Bcl-xl. In conclusion, these results suggest that the TFs from R. laevigata Michx fruit show good effects against H2O2-induced oxidative injury in PC12 cells by adjusting oxidative stress, and suppression of apoptosis and inflammation, and could be developed as a potential candidate to prevent oxidative stress in the future.
The effect of the total saponins from Rosa laevigata Michx fruit (RLTS) against acetaminophen (APAP)-induced liver damage in mice was evaluated in the present paper. The results showed that RLTS markedly improved the levels of liver SOD, CAT, GSH, GSH-Px, MDA, NO and iNOS, and the activities of serum ALT and AST caused by APAP. Further research confirmed that RLTS prevented fragmentation of DNA and mitochondrial ultrastructural alterations based on TdT-mediated dUTP nick end labeling (TUNEL) and transmission electron microscopy (TEM) assays. In addition, RLTS decreased the gene or protein expressions of cytochrome P450 (CYP2E1), pro-inflammatory mediators (IL-1β, IL-4, IL-6, TNF-α, iNOS, Bax, HMGB-1 and COX-2), pro-inflammatory transcription factors (NF-κB and AP-1), pro-apoptotic proteins (cytochrome C, p53, caspase-3, caspase-9, p-JNK, p-p38 and p-ERK), and increased the protein expressions of Bcl-2 and Bcl-xL. Moreover, the gene expression of IL-10, and the proteins including LC3, Beclin-1 and Atg5 induced by APAP were even more augmented by the extract. These results demonstrate that RLTS has hepatoprotective effects through antioxidative action, induction of autophagy, and suppression of inflammation and apoptosis, and could be developed as a potential candidate to treat APAP-induced liver damage in the future.
Matrix metalloproteinases (MMP) play a pivotal role in the pathogenesis of cardiovascular diseases. Their expressions are altered in response to a variety of stimuli, including growth factors, inflammatory markers, and cytokines. In this study, we demonstrated that platelet-derived growth factor-BB (PDGF-BB) induces a dose- and time-dependent increase in MMP-2 expression in rat vascular smooth muscle cells (VSMC). Treatment with either the Rho-associated protein kinase (ROCK) inhibitor Y-27632 or suppression of ROCK-1/2 by small interfering RNA technology significantly reduced the MMP-2 expression, thus suggesting that ROCK regulates such expression. Similar results were observed when VSMC were pretreated with either U0126 or SB203580, which are selective inhibitors of extracellular signal-regulated kinase and p38 mitogen-activated protein kinase, respectively, thus suggesting that these kinases are important for the induction of MMP-2 expression by PDGF-BB. In conclusion, these results described a novel mechanism in atherosclerosis through PDGF-BB signaling in VSMC, in which MMP-2 expression is induced via extracellular signal-regulated kinases and p38 mitogen-activated protein kinase phosphorylation, as well as ROCK.
The mechanism of blue light-induced retinal ganglion cell (RGC) injury is poorly understood. In this study, we established a patented light-emitting diode-based system to study the effects of long-term blue light exposure under culture conditions on RGC-5 cells. Long-term blue light exposure significantly reduced cell viability in a time-dependent manner and induced apoptosis and necrosis in RGC-5 cells. Long-term blue light exposure marked an increase in the expression of Bax and active Caspase-3 (p17), which was accompanied by Bcl-2 down-regulation, and displayed features of the mitochondria-dependent apoptosis pathway. Blue light exposure also increased the generation of reactive oxygen species (ROS), and was a strong inducer of ROS-sensitive protein nuclear factor erythroid 2-related factor 2 (Nrf2) and heme oxygenase-1 (HO-1) expression. Moreover, blue light exposure constitutively activated p38 mitogen-activated protein kinases and c-Jun NH2-terminal kinase (JNK), as well as induced the phosphorylation of extracellular signal-regulated kinase in the early phase, in blue light-exposed RGC-5 cells. The protein expression of c-jun and c-fos was further enhanced after RGC-5 cells were exposed to blue light. Taken together, these findings indicated that blue light induced RGC-5 cell line death in dependence upon exposure duration. The potential mechanisms for this phenomenon might be via activated mitochondria-dependent apoptosis, increased ROS production and protein expressions of Nrf2 and HO-1, and activated JNK/p38 MAPK signaling pathways.
Dentin, the predominant mineralized tissue of the tooth, comprises an extracellular matrix of collagen and a heterogeneous mixture of non-collagenous components, many of which have cellular signaling properties. These properties may be important in signaling stem cell involvement in tissue regeneration following injury and the present study investigates their morphogenic effects on differentiation of Bone Marrow Stromal Stem Cells (BMMSCs) in vitro. Non-collagenous dentin matrix proteins (DMPs) were isolated from healthy human teeth and their effects on BMMSCs behavior examined during in vitro culture. In vitro, DMPs enhanced alkaline phosphatase activity and mineralization in BMMSCs cultures as well as increasing the expression of dentinogenic and osteogenic differentiation markers (including runt-related transcription factor 2, osterix, bone sialoprotein, dentin sialophosphoprotein and osteocalcin) at both transcript and protein levels, with 10 μg/mL DMPs being the optimal stimulatory concentration. Expression of phosphor-ERK/phosphor-P38 in BMMSCs was up-regulated by DMPs and, in the presence of the ERK1/2- and p38-specific inhibitors, the differentiation of BMMSCs was inhibited. These data indicate that DMPs promote the dentinogenic/osteogenic differentiation of BMMSCs via the ERK/p38 MAPK pathways.
The present study was to investigate the effects and possible mechanisms of the total saponins from Dioscorea nipponica Makino (TSDN) against CCl₄-induced hepato-toxicity in mice. The mice were orally administrated with TSDN for seven days and then given CCl₄ (0.3%, 10 ml/kg i.p.). The results showed that TSDN significantly attenuated the activities of ALT and AST, consistent with hematoxylin-eosin staining. The ALP levels and relative liver weight were significantly decreased by TSDN compared with model group. Moreover, TSDN dramatically decreased MDA, iNOS and NO levels, while the levels of GSH, GSH-Px and SOD were increased. Further investigations showed that TSDN inhibited CCl₄-induced metabolic activation and CYP2E1 expression, down-regulated the levels of MAPKs phosphorylation, NF-κB, HMGB1, COX-2 as well as effectively suppressed the expressions of Caspase-3, Caspase-9, PARP and Bak. Quantitative real-time PCR assay demonstrated that TSDN obviously decreased the gene expressions of TNF-a, IL-1β, IL-6, IL-10, Fas, FasL, Bax as well as modulated Bcl-2 mRNA level. This is the first time to report the protective actions of the TSDN against CCl₄-induced liver damage in mice through suppression of inflammation and apoptosis. This natural product should be developed as a new drug for treatment of liver injury in future.
Apoptosis of microvascular endothelial cells plays a crucial role in the progression of various lung diseases and triggers microcirculatory disorder and organ dysfunction. LPS, an outer membrane component of Gram-negative bacteria, is one of the major virulence factors for lung diseases. Recent studies have shown that the Rho/Rho kinase (ROCK) pathway plays an important role in the regulation of apoptosis, inflammatory cell migration and chemokine production in various cell types and animal models. We therefore undertake this study to investigate the inhibitory effect of fasudil, a potent and selective inhibitor of ROCK, on LPS-induced apoptosis of rat pulmonary microvascular endothelial cells (PMVECs). The results suggested that fasudil effectively prevented LPS-induced injury of rat PMVECs, as determined by MTT assay, LDH activity assay, apoptosis and western blot analysis of apoptosis-related proteins Bcl-2 and Bax. Furthermore, the mechanisms underlying the protective effect were evaluated. We found that LPS-induced MYPT-1 phosphorylation was markedly suppressed by fasudil. Moreover, fasudil pretreatment obviously inhibited the activation of JNK and p38 MAPKs induced by LPS, whereas that of ERK1/2 was not affected by fasudil. In addition, inhibiting the JNK and p38 pathways by SP600125 and SB203580 respectively attenuated the LPS-induced apoptosis and regulated the expression of apoptosis-related proteins Bcl-2 and Bax. Taken together, these results demonstrate that fasudil exerts an anti-apoptotic effect in rat PMVECs, which is mediated by the inhibition of Rho/ROCK and its downstream JNK and p38 MAPKs.
Multiple studies have indicated that selective cyclooxygenase-2 (COX-2) inhibitors possess clinically chemopreventive and preclinically anticancer activities. Their long-term use, however, may be limited by the cardiovascular toxicity. This study tried to investigate whether an apple oligogalactan (AOG) could enhance the growth inhibitory effect of celecoxib on colorectal cancer. Caco-2 and HT-29 cell lines were exposed to different concentrations of AOG (0-1 g/L), celecoxib (0-25 μmol/L), and their combination. COX-2 levels were assessed by reverse transcription PCR and Western blot. COX-2 activity was evaluated by measuring prostaglandin E2 concentration. A colitis-associated colorectal cancer (CACC) mouse model was used to determine the effect of the combination in vivo. AOG (0.1-0.5 g/L) could potentiate the inhibitory effect of physiologic doses of celecoxib (5 μmol/L) on cell growth and decrease COX-2 expressions both at RNA and protein levels. In vivo, the combination (2.5% AOG plus 0.04% celecoxib, w/w) prevented against CACC in mice effectively. Our data indicate that AOG could potentiate the growth inhibitory effect of celecoxib on colorectal cancer both in vitro and in vivo through influencing the expression and function of COX-2 and phosphorylation of MAPKs, which suggests a new possible combinatorial strategy in colorectal cancer therapy.
Alfa-calendic acid and β-calendic acid, geometric and positional isomers of linolenic acid were previously shown to possess potent anticancer properties. In this study, we found that α-calendic acid and β-calendic acid could induce apoptosis and suppress invasion of human choriocarcinoma JEG-3 cells in vitro. Treatment with α-calendic acid and β-calendic acid significantly increased oxidative stress in human choriocarcinoma JEG-3 cells detected by the level of reactive oxygen species (ROS), lipid peroxidation production malondialdehyde (MDA), glutathione (GSH) and the effects of antioxidants NAC and α-tocopherol. Furthermore, oxidative stress activated the phosphorylation of p38MAPK. SB203580, a selective p38MAPK inhibitor, blocked the apoptosis induced by α-calendic acid and β-calendic acid by upregulating Bcl-2/Bax ratio and inhibition of the activation of Caspase-3 and Caspase-9. SB20350 also partially abrogated the cell invasion effects of α-calendic acid and β-calendic acid. These results suggested that α-calendic acid and β-calendic acid induced apoptosis and inhibited invasion in JEG-3 cells by activation of oxidative stress pathways and subsequent activation of P38MAPK.
The neuroprotective effect and mechanism of the flavonoid-rich extract (FRE) from Rosa laevigata Michx fruit on cerebral ischemia-reperfusion (I/R) injury were investigated. The contents of flavonoids, saponins and tannin were determined, and ten chemicals including chlorogenic acid, 4-hydroxy-3-methoxybenzoic acid, apigenin, luteolin, kaempferol, querce-tin, kaempferide-3-O-glucoside, quercetin-3-rhamnoside, rutin and isorhamnetin-3-O-β-rutinoside from the crude extract were separated. Oral administration of FRE obviously improved the survival rate and prevented I/R-induced disability and histological damage. Further works showed that the natural product had excellent antioxidant activity, significantly decreased DNA fragmentation, up-regulated the expression of Bcl-2, and down-regulated the expressions of p53, Apaf1, Fas, FasL, Bax, Bid, cytochrome C and active Caspase-3, -9 and -8. Moreover, the FRE decreased the expressions of NF-κB, iNOS, MMP-9, COX-2, TNF-α, IL-1β, IL-4, IL-6, and down-regulated the levels of p-JNK, p-ERK and p-p38 in MAPK pathways. Therefore, the flavonoid-rich extract from R. laevigata Michx fruit has the potential actions for treatment of ischemic stroke due to its anti-oxidant, anti-apoptosis and anti-inflammatory properties.
MicroRNAs have tumor suppressive or oncogenic roles in carcinogenesis. This study aimed to investigate the mechanism of let-7c in suppressing lung cancer cell proliferation. First, let-7c was revealed to be able to inhibit lung adenocarcinoma cell proliferation significantly. TRIB2 was further demonstrated to be a novel target and negatively regulated by let-7c. As downstream signals of TRIB2, the activities of C/EBP-α and phosphorylated p38MAPK were increased obviously in let-7c-treated cells compared with controls. Our results demonstrate that, through regulating the expression of TRIB2 and its downstream factors, let-7c can effectively inhibit A549 cell proliferation in vitro and in vivo.
Urocortin (UCN1) is a member of corticotrophin-releasing factor (CRF) family, which has been proven to participate in inflammation. Previous work showed that dihydrotestosterone (DHT) could promote the inflammatory process. Little is known about the effect of DHT on UCN1 expression. The aim of our study is to investigate the effects and underlying mechanisms of DHT on endothelial UCN1 expression in the absence and presence of induced inflammation. Therefore, we tested the alterations of endothelial UCN1 expression treated with DHT in the presence or absence of lipopolysaccharide (LPS). Our data showed that DHT alone decreased UCN1 levels, which were attenuated in the presence of the androgen receptor (AR) antagonist flutamide. Conversely, in the presence of LPS, DHT augmented the LPS-induced increase in UCN1 expression, which was, interestingly, not affected by flutamide. When cells were treated with DHT alone, AR was upregulated and translocated into the nuclei, which might repress UCN1 expression via a potential androgen-responsive element found in human CRF family promoter. In the presence of LPS, DHT did not influence AR expression and location while it increased toll-like receptor 4 expression and activation, which was not altered by flutamide. DHT enhanced LPS-induced p38MAPK, ERK1/2, and nuclear factor κB pathway activation, which may contribute to the elevated expression of UCN1. These data suggest that DHT differentially influences UCN1 levels under normal and inflammatory conditions in human umbilical vein endothelial cells, which involves AR-dependent and -independent mechanisms respectively.
It has been well documented in in vitro studies that ambient airborne particulate matter (PM) with an aerodynamic diameter less than 2.5 μm (PM(2.5)) is capable of inducing oxidative stress, which plays a key role in PM(2.5)-mediated cytotoxicity. Although nuclear factor erythroid-2-related factor 2 (Nrf2) has been shown to regulate the intracellular defense mechanisms against oxidative stress, a potential of the Nrf2-mediated cellular defense against oxidative stress induced by PM(2.5) remains to be determined. This study was aimed to explore the potential signaling pathway of Nrf2-mediated defense mechanisms against PM(2.5)-induced oxidative stress in human type II alveolar epithelial A549 cells. We exposed A549 cells to PM(2.5) particles collected from Beijing at a concentration of 16 μg/cm(2). We observed that PM(2.5) triggered an increase of intracellular reactive oxygen species (ROS) in a time-dependent manner during a period of 2 h exposure. We also found that Nrf2 overexpression suppressed and Nrf2 knockdown increased PM(2.5)-induced ROS generation. Using Western blot and confocal microscopy, we found that PM(2.5) exposure triggered significant translocation of Nrf2 into nucleus, resulting in AKT phosphorylation and significant transcription of ARE-driven phases II enzyme genes, such as NAD(P)H:quinone oxidoreductase (NQO-1), heme oxygenase-1 (HO-1), and glutamate-cysteine ligase catalytic subunit (GCLC) in A549 cells. Evaluation of signaling pathways showed that a phosphatidylinositol 3-kinase (PI3K) inhibitor (LY294002), but not an ERK 1/2 inhibitor (PD98059) or a p38 MAPK (SB203580), significantly down-regulated PM(2.5)-induced Nrf2 nuclear translocation and HO-1 mRNA expression, indicating PI3K/AKT is involved in the signaling pathway leads to the PM(2.5)-induced nuclear translocation of Nrf2 and subsequent Nrf2-mediated HO-1 transcription. Taken together, our results suggest that PM(2.5)-induced ROS may function as signaling molecules to activate Nrf2-mediated defenses, such as HO-1 expression, against oxidative stress induced by PM(2.5) through the PI3K/AKT signaling pathway.
Formyl peptide receptor 1 (FPR1) plays an important role in the rapid progression of glioblastoma and has been considered as a molecular target for the treatment. Previously, we have shown that oligomer proanthocyanidins (F2, degree of polymerization 2-15), isolated from grape seeds, inhibited FPR1-mediated chemotaxis of U-87 glioblastoma cells. In the present study, we investigated the capacity of F2 to interact with FPR1. The cross attenuation of chemotaxis revealed that F2 shared FPR1 with formyl-methionyl-leucyl-phenylalanine (fMLF), which is a prototype agonist of FPR1. F2 was chemotactic for U-87 cells, and the chemotactic response was abolished when FPR1 gene was silenced or FPR1 was competitively occupied. We further show that F2 specifically blocked the binding of fluorescent agonist to FPR1. Interestingly, F2 exhibited the characteristic of a partial agonist for FPR1, as shown by its capacity to activate FPR1-mediated PI3K-PKC-MAPK pathways. Meanwhile, F2 also attenuated fMLF-triggered MAPK activation, suggesting that F2 could antagonize the effect of an agonist. Furthermore, F2 abolished the invasion of U-87 cells induced by fMLF. Thus, we have identified F2 as a novel, partial agonist for FPR1, which may be useful for glioblastoma therapy.
Interferon regulatory factor (IRF) 3, a member of the highly conserved IRF family transcription factors, plays a pivotal role in innate immune response, apoptosis, and oncogenesis. Recent studies have implicated IRF3 in a wide range of host defense. However, whether IRF3 induces defensive responses to hypertrophic stresses such as biomechanical stress and neurohumoral factors remains unclear. Herein, we employed an IRF3-deficient mouse model, cardiac-specific IRF3-overexpression mouse model and isolated cardiomyocytes to investigate the role of IRF3 in cardiac hypertrophy induced by aortic banding (AB) or isoproterenol (ISO). The extent of cardiac hypertrophy was quantitated by echocardiography as well as by pathological and molecular analysis. Our results demonstrate that IRF3 deficiency profoundly exacerbated cardiac hypertrophy, whereas overexpression of IRF3 in the heart significantly blunted pathological cardiac remodeling induced by pressure overload. Similar results were also observed in cultured cardiomyocytes upon the treatment with ISO. Mechanistically, we discovered that IRF3 interacted with ERK2 and thereby inhibited the ERK1/2 signaling. Furthermore, inactivation of ERK1/2 by U0126 offset the IRF3-deficient-mediated hypertrophic response induced by aortic banding. Altogether, these data demonstrate that IRF3 plays a protective role in AB-induced hypertrophic response by inactivating ERK1/2 in the heart. Therefore, IRF3 could be a new target for the prevention and therapy of cardiac hypertrophy and failure.
Diosgenin (Dio), a major active component of steroidal sapogenin of the traditional Chinese herb Dioscorea zingiberensis C.H.Wright, shows various activities including anti-inflammatory, anti-thrombotic activities, anti-cancer properties etc. In the present study, we found that diosgenin significantly suppressed the phosphorylation of lung NF-κB p50/p65 and MAPK/p38 in lipopolysaccharide (LPS)-induced acute lung injury (ALI) in mice, when given orally at doses of 0.1, 1.0 and 10mg/kg 1h prior to LPS challenge (30 mg/kg, intravenous injection). Moreover, diosgenin attenuated the lung histopathological changes such as pulmonary edema, coagulation and infiltration of inflammatory cells. In addition, diosgenin significantly decreased the lung wet to dry weight (W/D) ratio and nitrite/nitrate content at three doses, and also markedly inhibited LPS-induced body temperature decrease and nitrite/nitrate elevation in plasma. Besides, diosgenin could significantly suppress activation of NF-κB p65/p50, p38 and expression of inducible nitric oxide synthase (iNOS) in LPS-induced THP-1 cells. Our findings indicate the potential application of diosgenin for ALI treatment.
The adhesion of monocytes to activated vascular endothelial cells is a critical event in the initiation of atherosclerosis. Adhesion is mediated by oxidized low-density lipoprotein (ox-LDL) which up-regulates inflammatory markers on endothelial cells. Here we report that (±) 7, 8-dihydroxy-3-methyl-isochromanone-4 (XJP-1), an inhibitor of ox-LDL-induced adhesion of monocytes to endothelial cells blocks cellular functions which are associated with adhesion. We show that XJP-1 down-regulates ox-LDL-induced over-expression of adhesion molecules (ICAM-1 and VCAM-1) in a dose-dependent manner in human umbilical vein endothelial cells (HUVECs), attenuates ox-LDL-induced up-regulation of low-density lipoprotein receptor (LOX)-1, decreases generation of reactive oxygen species (ROS), blocks translocation of nuclear factor-kappa B (NF-κB) activity, and prevents activation of c-Jun N-terminal kinase (JNK)/p38 pathways in endothelial cells. These findings suggest that XJP-1 may attenuate ox-LDL-induced endothelial adhesion of monocytes by blocking expression of adhesion molecules through suppressing ROS/NF-κB, JNK and p38 pathways.
Keloid is a specific skin scar that expands beyond the boundaries of the original injury as it heals. The invasive nature of keloid and notable migratory activity of fibroblasts are a hallmark, which distinguishes keloids from other common scars. Madecassoside, a triterpenoid saponin occurring in Centella asiatica herbs, possesses unique pharmacological properties to enhance wound-healing and diminish keloid formation. However, the effects of madecassoside on the formation of keloid scars have been poorly understood. Here, we focused on the potential of madecassoside on the migration of keloid-derived fibroblasts (KFs) and its mechanism. Primary KF, originating from human earlobe keloids, were purified and cultured, and then treated with madecassoside (10, 30, and 100μM). In both transwell migration assays and scratch-wound-closure assays, KF migration was considerably suppressed by madecassoside pretreatment. Furthermore, KFs treated with madecassoside showed decreased F-actin filaments, as revealed by fluorescein isothiocyanate (FITC)-phalloidin staining and confocal microscopy. By Western blot analysis, madecassoside was shown to remarkably attenuate the phosphorylation of cofilin, p38 MAPK and phosphatidylinositol-3-kinase (PI3K)/AKT signaling, but only exhibited a minor effect on MMP-13 and little effect on ERK1/2 phosphorylation. It was concluded that madecassoside could be of great use in the treatment and/or prevention of hypertrophic scars and keloids.
Cellular FLICE-inhibitory protein (cFLIP) is a member of the tumour necrosis factor signalling pathway and a regulator of apoptosis, and it has a role in cardiac remodelling following myocardial infarction (MI) that remains largely uncharacterised. This study aimed to determine the function of cFLIP as a potential mediator of post-infarction cardiac remodelling. Our results show diminished cFLIP expression in failing human and murine post-infarction hearts. Genetically engineered cFLIP heterozygous (cFLIP+/-, HET) mice, cardiac-specific cFLIP-overexpressing transgenic (TG) mice and their respective wild-type (WT) and non-transgenic controls were subjected to MI by permanent ligation of their left anterior descending artery. Cardiac structure and function were assessed by echocardiography and pressure-volume loop analysis. Apoptosis, inflammation, angiogenesis, and fibrosis were evaluated in the myocardium. The HET mice showed exacerbated left ventricular (LV) contractile dysfunction, dilatation, and remodelling compared with WT mice 28 days after MI. Impaired LV function in the HET mice was associated with increases in infarct size, hypertrophy, apoptosis, inflammation, and interstitial fibrosis, and reduced capillary density. The TG mice displayed the opposite phenotype after MI. Moreover, adenovirus-mediated overexpression of cFLIP decreased LV dilatation and improved LV function and remodelling in both HET and WT mice. Further analysis of signalling events suggests that cFLIP promotes cardioprotection by interrupting JNK1/2 signalling and augmenting Akt signalling. In conclusion, our results indicate that cFLIP protects against the development of post-infarction cardiac remodelling. Thus, cFLIP gene delivery shows promise as a clinically powerful and novel therapeutic strategy for the treatment of heart failure after MI.
Praeruptorin C, D, and E (PC, PD, and PE) are three pyranocoumarins isolated from the dried root of Peucedanum praeruptorum Dunn of Umbelliferae. In the present study, we investigated the anti-inflammatory effect of these compounds in lipopolysaccharide (LPS)-stimulated RAW264.7 macrophage cells. Pyranocoumarins significantly inhibited LPS-induced production of nitric oxide, interleukin-6 (IL-6), and tumor necrosis factor-α (TNF-α). The mRNA and protein expressions of inducible nitric oxide synthase, IL-6, and TNF-α were also suppressed by these compounds. Both PD and PE exhibited greater anti-inflammatory activities than PC. Further study showed that pyranocoumarins suppressed the cytoplasmic loss of inhibitor κB-α protein and inhibited the translocation of NF-κB from cytoplasm to nucleus. In addition, pyranocoumarins suppressed LPS-induced STAT3 tyrosine phosphorylation. Taken together, the results suggest that pyranocoumarins may exert anti-inflammatory effects in LPS-stimulated RAW 264.7 macrophages through the inhibition of NF-κB and STAT3 activation.
We examined the effects of anti-six-transmembrane epithelial antigen of the prostate-4 (STEAP4) antibodies on glucose transport in mature adipocytes and determined the mechanism of insulin resistance in obesity. Western blotting was performed to determine STEAP4 expression, to assess translocation of insulin-sensitive glucose transporter 4 (GLUT4), and to measure phosphorylation and total protein content of insulin-signaling proteins. Confocal laser microscopy and flow cytometry were used to detect intracellular reactive oxygen species (ROS) and fluctuations in mitochondrial membrane potential (ΔΨ). ATP production was measured by using a luciferase-based luminescence assay kit. After the application of anti-STEAP4 antibodies at 0.002 mg/mL, adipocytes exhibited reduced insulin-stimulated glucose transport by attenuating the phosphorylation of IRS-1, PI3K (p85), and Akt. The antibodies also potentially increase the level of ROS and decrease cellular ATP production and ΔΨ. In conclusion, (i) STEAP4 regulates the function of IRS-1, PI3K, and Akt and decreases insulin-induced GLUT4 translocation and glucose uptake; (ii) ROS-related mitochondrial dysfunction may be related to a reduced IRS-1 correlation with the PI3K signaling pathway, leading to insulin resistance. These observations highlight the potential role of STEAP4 in glucose homeostasis and possibly in the pathophysiology of type 2 diabetes related to obesity and may provide new insights into the mechanisms of insulin resistance in obesity.
OBJECTIVES:
The aim of this study was to investigate effects of mineral trioxide aggregate (MTA) on odonto/osteogenic differentiation of bone marrow stromal cells (BMSCs) from craniofacial bones.
MATERIALS AND METHODS:
Craniofacial BMSCs were isolated from rat mandible and effects of MTA on their proliferation, differentiation and MAPK pathway involvement were subsequently investigated, in vitro. MTT (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl-2,5-tetrazoliumbromide) assay was performed to evaluate proliferation of the MTA-treated cells. Alkaline phosphatase (ALP) activity, alizarin red staining, real-time reverse transcription polymerase chain reaction and western blot assays were used to assess differentiation capacity as well as MAPK pathway involvement.
RESULTS:
0.02 mg/ml MTA-treated BMSCs had significantly higher ALP activity and formed more mineralized nodules than the untreated group. Odonto/osteoblastic marker genes/proteins (Alp, Runx2/RUNX2, Osx/OSX, Ocn/OCN and Dspp/DSP respectively) in MTA-treated cells were remarkably upregulated compared to untreated ones. Mechanistically, phosphorylated Jun N-terminal kinase (P-JNK) and phosphorylated extracellular regulated protein kinases (P-ERK) in MTA-treated BMSCs increased significantly in a time-dependent manner, while inhibition of JNK and ERK MAPK pathways dramatically blocked MTA-induced odonto/osteoblastic differentiation, as indicated by reduced ALP levels, weakened mineralization capacity and downregulated levels of odonto/osteoblastic marker genes (Alp, Runx2, Osx, Ocn and Dspp).
CONCLUSION:
Mineral trioxide aggregate promoted odonto/osteogenic capacity of craniofacial BMSCs via JNK and ERK MAPK signalling pathways.
© 2014 John Wiley & Sons Ltd.
SCOPE:
Flavonoids have well-known antioxidant, anti-inflammatory, and anti-cancer activities. Isoflavone genistein is considered a potent antioxidant agent against oxidative stress. Although several mechanisms have been proposed, a clear antioxidant mechanism of genistein is still remained to be answered.
METHODS AND RESULTS:
In this study, we focused on the concerted effects on expression of Nrf2 and phase II enzyme pathway components. Transient transfection assays, RT-PCR and immunoblot analysis were performed to study its molecular mechanisms of action. In Caco-2 cells, treatment with genistein markedly attenuated H(2)O(2) -induced peroxide formation; this amelioration was reversed by buthionine sulfoximine(GCLC inhibitor) and zinc protoporphyrin(HO-1 inhibitor). Genistein increased HO-1 and GCLC mRNA and protein expression. Genistein treatment activated the ERK1/2 and PKC signaling pathway; therefore increased Nrf2 mRNA and protein expression. The roles of the ERK1/2 and PKC signaling pathway were determined using PD98059 (ERK1/2 inhibitor) and GF109203X (PKC inhibitor) and RNA interference directed against Nrf2. Both inhibitors and siNrf2 abolished genistein-induced HO-1 and GCLC protein expression. These results suggest the involvement of ERK1/2, PKC, and Nrf2 in inducing HO-1 and GCLC by genistein.
CONCLUSION:
Our studies show that genistein up-regulated HO-1 and GCLC expression through the EKR1/2 and PKC /Nrf2 pathways during oxidative stress.
© 2012 WILEY-VCH Verlag GmbH & Co. KGaA, Weinheim.
BACKGROUND:
Riccardin D-26, a synthesized macrocyclic bisbibenzyl compound, might possess anti-cancer properties. We aimed to evaluate the efficacy of Riccardin D-26 as a candidate compound for treatment of cancers with sensitive or drug resistant cells.
METHODS:
Experiments were performed on human oral squamous carcinoma KB cells and vincristin-selected MDR KB/VCR cells. The inhibition of cell growth was evaluated by colorimetric and clonogenic assays. The apoptotic cells were determined by the Annexin V-FITC/PI staining assay. JC-1 fluorescence probe was used to examine the mitochondria membrane potential (MMP). Further experiments were performed in nude mice bearing KB or KB/VCR xenografts. Riccardin D-26 was administered by injection for 2weeks. The specimens of KB and KB/VCR xenografts were removed for TUNEL staining and Western blotting analysis.
RESULTS:
Riccardin D-26 significantly inhibited cancer growth in both KB and KB/VCR cells. Riccardin D-26's activity in cancer cells was greater than that in human normal liver cells. In mice, Riccardin D-26 effectively prevented the growth of KB and KB/VCR xenografts without significant toxicity. Further studies suggested that Riccardin D-26 inhibited cancer growth by inducing apoptosis in the activation of mitochondria-mediated intrinsic apoptosis pathway. Riccardin D-26 also possessed this activity in regulation of mitogen-related protein kinases such as MAPK and PI3K/Akt, which is associated with its inhibitory effect on KB/VCR cells.
CONCLUSIONS:
Riccardin D-26 possessed an anti-proliferation activity against both sensitive KB and MDR KB/VCR cancer cells.
GENERAL SIGNIFICANCE:
Riccardin D-26 could be a promising agent for treatment of cancers with sensitive or drug resistant cells.
Copyright © 2012 Elsevier B.V. All rights reserved.
ETHNOPHARMACOLOGICAL RELEVANCE:
Oxymatrine is extracted from the traditional Chinese herb Sophora flavescens Ait, possesses anti-inflammatory, anti-oxidative and anti-apoptotic properties, and has been used for the treatment of chronic viral hepatitis and many other diseases.
AIMS OF THE STUDY:
This study aimed to investigate the effects of oxymatrine on inflammatory response mediated by Toll-like receptor4 (TLR4) and nuclear factor kappa-B (NF-κB), oxidative injury induced by 12/15 lipoxygenase (12/15-LOX), phosphorylated p38 mitogen activated protein kinase (phosphor-p38 MAPK) and cytosolic phospholipase A2 (cPLA2), and neuronal cell apoptosis in rat brain with intracerebral hemorrhage (ICH).
MATERIALS AND METHODS:
Wistar rats were treated intraperitoneally with 60 or 120mg/kg of oxymatrine daily for 5 days following ICH. The rats were sacrificed at hour 2, 6, 12, 24, 48, 72, and 120 after ICH. The gene expressions of TLR-4 and NF-κB, the levels of TNF-alpha, interleukin-1beta, interleukin-6, 12/15-LOX, phospho-p38 MAPK and cPLA2, and the number of apoptotic neuronal cells in rat brain were determined.
RESULTS:
Oxymatrine at 120mg/kg significantly suppressed gene expressions of TLR-4 and NF-κB, decreased levels of TNF-alpha, interleukin-1beta and interleukin-6, inhibited synthesis of 12/15-LOX, phospho-p38 MAPK and cPLA2 protein, and mitigated apoptotic neuronal changes following ICH in rat.
CONCLUSION:
Oxymatrine at 120mg/kg following ICH inhibits inflammatory responses, oxidative injury, and neuronal cell apoptosis in rats.
Copyright © 2012 Elsevier Ireland Ltd. All rights reserved.
AIM:
We investigated whether 8-dihydroxy-3-methyl-isochromanone (XJP-1), a novel angiotensin-converting enzyme inhibitor (ACEI), exhibited inhibitory activity to lipopolysaccharide (LPS)-accelerated vascular inflammation.
METHODS:
Human umbilical vein endothelial cells (HUVECs) were isolated from human umbilical cords and cultured. The direct effect of XJP-1 on the activation of endothelial cells was measured using MTT assay. Nitric oxide (NO) in the culture medium was measured using Griess method. The expression of cell adhesion molecules (ICAM-1 and VCAM-1) was determined by flow cytometry and RT-PCR. The protein expression levels of tumor necrosis factor-α (TNF-α), monocyte chemotactic protein (MCP)-1, and endothelin-1 (ET-1) secretion were measured using ELISA. Quantitative analysis of eNOS, iNOS, inhibitory factor NF-κB (IκB) and MAPKs were determined using Western blot analysis. The translocation of NF-κB from the cytoplasm to the nucleus was determined using immunofluorescence.
RESULTS:
XJP-1 significantly inhibited LPS-mediated endothelial cell dysfunction, as measured by NO production, iNOS expression, adhesion molecule (ICAM-1, VCAM-1) expression, and chemokine (TNF-α, MCP-1) production in vitro. It up-regulated eNOS expression in the same experimental setting. XJP-1 alone was found non-cytotoxic at the concentration up to 1000μM. The mechanistic investigations of XJP-1 suppression LPS-induced inflammation in HUVECs revealed that XJP-1 blocked NF-κB nuclear entry in an IκB-dependent manner, as well as inhibited MAPK activation induced by LPS. XJP-1 reduced endothelin-1 secretion and increased nitric oxide metabolite production by HUVECs. However, the effect of XJP-1 on nitric oxide and endothelin-1 metabolite production is mediated by the activation of bradykinin B(2) receptor being counteracted, at least in part, by a specific antagonist.
CONCLUSION:
XJP-1 inhibited LPS-induced cytotoxicity and inflammatory response. The mechanism underlying this protective effect might be related to the inhibition of MAPK and NF-κB signaling pathway activation, suggesting the potential inhibition of the atherosclerotic process by suppressing the expression of chemoattractant molecules and monocyte adhesion. XJP-1 also has an effect in improving endothelin-1 through activating bradykinin B(2) receptor. These findings indicated that XJP-1 is potentially a novel therapeutic candidate for the treatment of atherosclerosis.
Copyright © 2011 Elsevier Ireland Ltd. All rights reserved.
![Western Blot - Anti-p38 MAPK (phospho Thr180 + Tyr182) Antibody [ARC51018] (A307918) - Antibodies.com](https://cdn.antibodies.com/image/catalog/307/A307918_1.jpg?profile=product_alternative)
![Western Blot - Anti-p38 MAPK (phospho Thr180 + Tyr182) Antibody [RM243] (A121194) - Antibodies.com](https://cdn.antibodies.com/image/catalog/121/A121194_1.png?profile=product_alternative)
![Western Blot - Anti-p38 MAPK Antibody [RM245] (A121383) - Antibodies.com](https://cdn.antibodies.com/image/catalog/121/A121383_1.png?profile=product_alternative)
![Immunohistochemistry - Anti-MAPK14 Antibody [CPTC-MAPK14-1] (A248270) - Antibodies.com](https://cdn.antibodies.com/image/catalog/248/A248270_1.jpg?profile=product_alternative)
![Immunohistochemistry - Anti-MAPK14 Antibody [CPTC-MAPK14-1] - BSA and Azide free (A251452) - Antibodies.com](https://cdn.antibodies.com/image/catalog/251/A251452_1.jpg?profile=product_alternative)



![Western Blot - Anti-p38 MAPK (phospho Thr180 + Tyr182) Antibody [ARC51018] (A307918) - Antibodies.com](https://cdn.antibodies.com/image/catalog/307/A307918_1.jpg?profile=product_alternative)
![Western Blot - Anti-p38 MAPK (phospho Thr180 + Tyr182) Antibody [RM243] (A121194) - Antibodies.com](https://cdn.antibodies.com/image/catalog/121/A121194_1.png?profile=product_alternative)
![Western Blot - Anti-p38 MAPK Antibody [RM245] (A121383) - Antibodies.com](https://cdn.antibodies.com/image/catalog/121/A121383_1.png?profile=product_alternative)


![Immunohistochemistry - Anti-MAPK14 Antibody [CPTC-MAPK14-1] (A248270) - Antibodies.com](https://cdn.antibodies.com/image/catalog/248/A248270_1.jpg?profile=product_alternative)
![Immunohistochemistry - Anti-MAPK14 Antibody [CPTC-MAPK14-1] - BSA and Azide free (A251452) - Antibodies.com](https://cdn.antibodies.com/image/catalog/251/A251452_1.jpg?profile=product_alternative)