Mark F Rosenberg, PhD | 21st May 2025
Neural crest cells differentiate into melanocytes during embryonic development. These specialized cells utilize their melanosome organelles to produce melanin pigments that color skin, hair, and eyes while protecting from UV radiation.
Melanocytes can be identified through characteristic molecular markers using methods such as Immunohistochemistry (IHC), Immunofluorescence (IF) and Flow cytometry (FC). These techniques enable researchers to investigate melanocyte development and function while detecting melanocytic disorders, particularly melanomas.
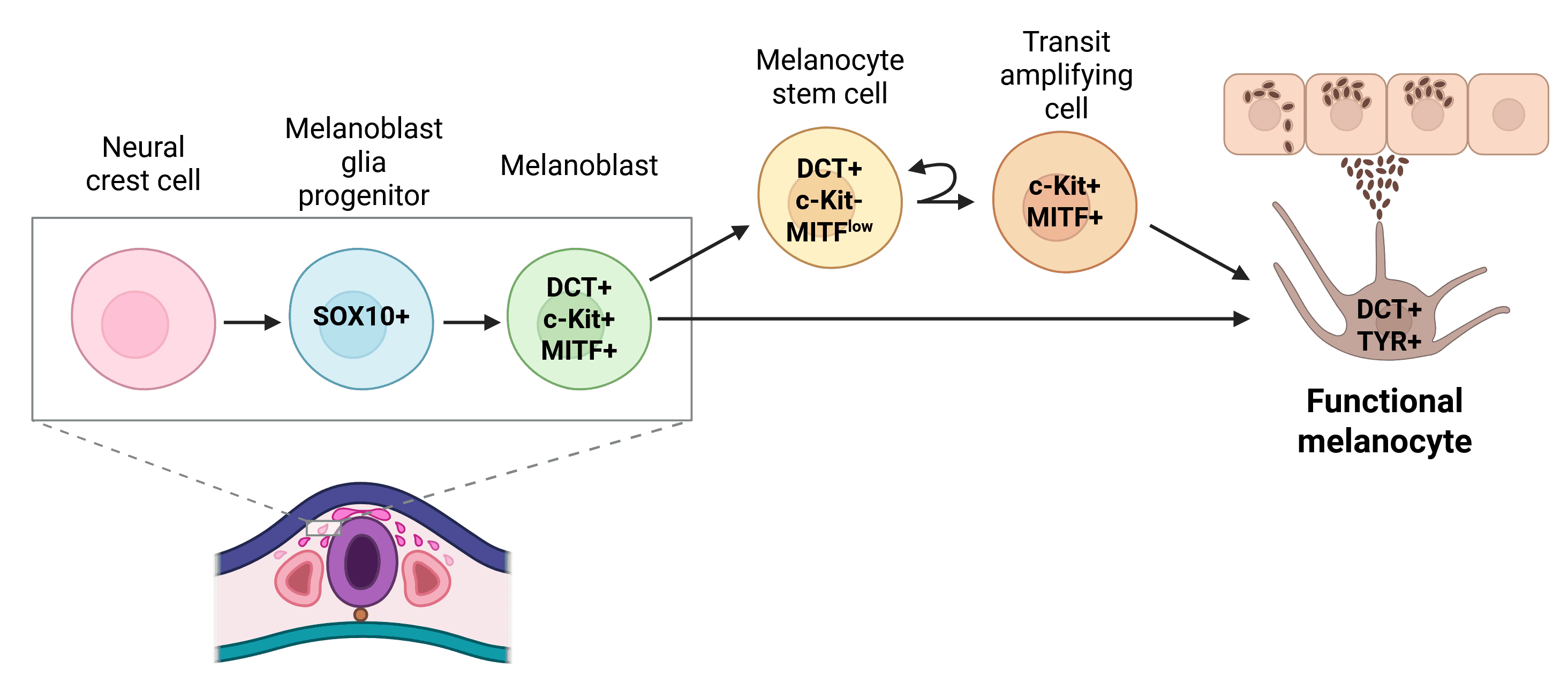
Melanocytes originate from neural crest cells between the neural plate and non-neural ectoderm. These cells undergo epithelial-to-mesenchymal transition, migrate through the embryo, and differentiate into melanocytes. Key regulatory proteins guide this process and serve as markers for tracking melanocyte development (Figure 1).1
Figure 1: Mammalian melanocyte development. In mammals, melanoblasts differentiate from neural crest cells (NCCs). Melanocyte stem cells are able to replenish functional melanocytes via rapidly proliferating transit-amplifying cells. Key markers are indicated within the cells.
In the skin, mature melanocytes reside in the basal epidermis layer. Dendritic extensions on melanocytes transport melanin-filled melanosomes to adjacent keratinocytes,2 aiding in protecting skin cells from UV radiation. Melanocytes synthesize two types of melanin within melanosomes: eumelanin (brown/black) and pheomelanin (yellow/red).3
The proteins involved in the production and movement of melanin, especially tyrosinase, are reliable markers for pinpointing melanocytes.
Melanocytes also populate eye tissue, inner ear, leptomeninges, and mucosal surfaces. Researchers must consider this anatomical distribution when picking markers, as expression patterns vary by location.4
MITF (Microphthalmia-associated Transcription Factor)
MITF works as the master regulator of melanocyte growth. This basic helix-loop-helix protein activates genes for melanin synthesis (TYR, TYRP1, DCT). It guides how melanocytes proliferate and differentiate. MITF expression is confined to melanocytic cells, facilitating melanoma identification (Figure 2). The MITF-M isoform is expressed exclusively in melanocytes, enabling distinction from other MITF-expressing cell types.5 MITF is found in cell nuclei, making it particularly useful for distinguishing melanocytes by IHC.
SOX10 (SRY-Box Transcription Factor 10)
SOX10 is expressed in neural crest cells and persists in melanocytes as they migrate; this protein regulates MITF expression and activates melanocyte-specific genes.6 SOX10 reliably detects amelanotic melanocytes and detects melanoma subtypes better than other markers, especially the desmoplastic and spindle cell variants.7 However, SOX10 appears in Schwann cells and other neural crest derivatives, so researchers must interpret their results carefully.
PAX3 (Paired Box 3)
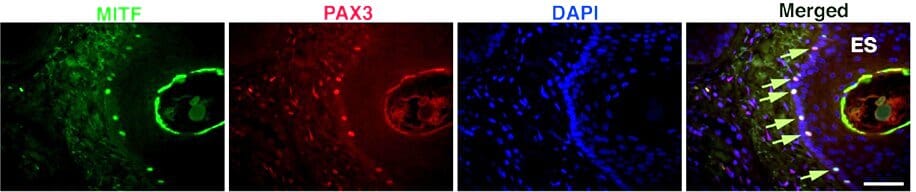
PAX3 also functions upstream of MITF and helps melanocytes proliferate. This protein is expressed early in melanoblasts and persists in mature melanocytes (Figure 2). Researchers use PAX3 antibodies to trace melanocyte precursors and explore how these cells differentiate.8 PAX3 is not as specific as MITF or SOX10 for marking mature melanocytes because it is expressed in muscle satellite cells and some neurons.
Figure 2: MITF and PAX3 expression in skin melanocytes. Arrows indicate co-expression. Edited and reproduced under CC BY 4.0 from Eccles et al., Front. Oncol. 3 (2013).
Melanosomes are organelles inside melanocytes that synthesize and store melanin. These structures house protein markers that correlate with pigmentation amounts. They comprise enzymes (tyrosinase) and structural proteins (PMEL, MART-1).
Tyrosinase
Tyrosinase catalyzes the conversion of tyrosine to dopaquinone, and helps its polymerization into melanin.9 Antibodies that bind this marker enzyme pinpoint melanocytic lineage cells, creating a distinctive granular cytoplasmic staining in IHC. The amounts of tyrosinase correlate with melanocyte differentiation state, making it valuable for assessing cellular maturation.2
TYRP1 (Tyrosinase-Related Protein 1) and DCT/TYRP2 (Dopachrome Tautomerase)
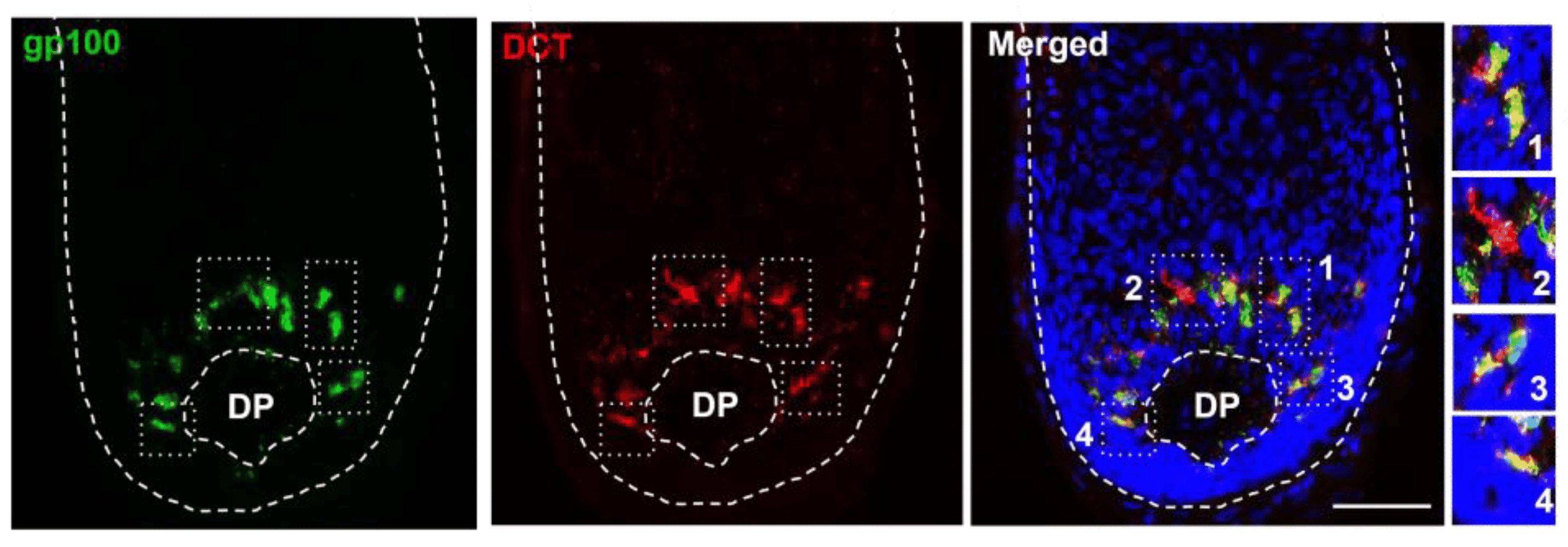
TYRP1 and DCT/TYRP2 work downstream of tyrosinase in the melanin synthesis pathway. TYRP1 antibodies stain these proteins and track melanin synthesis (Figure 3). DCT is expressed earlier in development, enabling detection of melanocyte precursors before they make melanin.10
PMEL (Premelanosome Protein)
PMEL, also known as gp100 or Silver, forms the fibrillar matrix where melanin is deposited during melanosome maturation.11 The HMB-45 antibody recognizes PMEL, an essential marker for melanocytic lesions (Figure 3). HMB-45 produces granular cytoplasmic staining in IHC, correlating with melanocyte differentiation and helping classify melanocytic lesions.12
Figure 3: PMEL and DCT expression in human scalp. Immunofluorescence of gp100 and DCT in the adult human scalp anagen hair bulb, showing some subpopulations that express only one of these markers. DP, dermal papilla. White boxes inset correspond to zoomed images on right. Edited and reproduced under CC BY 4.0 from Casalou et al., Int. J. Mol. Sci. 24, 12809 (2023).
MART-1/Melan-A
MART-1/Melan-A is a melanosomal transport protein essential for melanosome formation. Clinicians use the A103 antibody against Melan-A to find melanocytic lesions.13 Melan-A staining appears throughout the cytoplasm and remains effective even in amelanotic lesions. However, it also appears in adrenal cortical cells, so the results need careful review.
These markers help define melanocytes in flow cytometry and live cell studies.
c-Kit (CD117)
c-Kit is a receptor tyrosine kinase necessary for cell proliferation, survival, and migration. Mutations in the c-Kit gene cause piebaldism, characterized by patches of depigmented skin.14 Mature melanocytes keep expressing c-Kit, which helps find these cells. c-Kit appears at the cell membranes in tissue samples following staining, consistent with its role as a transmembrane receptor.
p75 Neurotrophin Receptor (CD271)
Neural crest cells express p75NTR during development, which remains in adult melanocytes. This receptor identifies melanocyte stem cells and early melanoma cells.15 SOX10 controls how much p75NTR is made, with more in melanocyte precursors than in mature cells. This helps find early melanocyte lineage cells and melanoma subpopulations with stem cell-like properties.
S100 proteins (S100B)
The S100 family of calcium-binding proteins are found in the cytoplasm and nuclei of neural crest derivatives, Schwann cells and some neurons.16 Due to this diverse expression, antibodies staining this protein are not as specific as other melanocyte markers and often need to be combined with other markers. S100 can detect desmoplastic melanomas and exhibits nuclear and cytoplasmic staining, facilitating studies of protein function.
Nestin
Nestin, an intermediate filament protein, is expressed in melanocyte stem cells in the hair follicle bulge region. Nestin antibodies help understand melanocyte regeneration and homeostasis.17 However, markers for Nestin are not specific enough and require co-staining with other markers for reliable melanocyte identification.
Melanocyte markers play key roles in dermatopathology and oncology. They help clinicians identify melanocyte-derived conditions, from benign nevi (moles) to aggressive melanomas.
Melanoma Diagnostic Markers
Melanoma is a skin cancer that starts in melanocytes. Clinicians employ several markers to:
Standard marker panels include S100, HMB-45 (PMEL), and Melan-A/MART-1. S100 detects most melanomas but isn't specific. HMB-45 and Melan-A are more specific but miss some melanoma subtypes.18
SOX10 works well across all melanoma types, even with tough cases like desmoplastic melanomas that lack other markers.7 Recent studies show SOX10 has >95% sensitivity for desmoplastic melanoma compared to <80% for S100.19
Additional markers help in hard-to-diagnose cases. For example, p16 (CDKN2A) expression is not easy to detect in melanoma, in contrast to benign moles.20 Conversely, Ki-67, a proliferation marker, is expressed more in melanomas than in benign moles. This highlights the importance of strategic marker selection to improve sensitivity and diagnosis.21
Melanocyte markers also facilitate finding micrometastases in sentinel lymph nodes. SOX10, MART-1, or HMB-45 staining with IHC improves detection sensitivity compared to standard testing, influencing melanoma staging and therapeutic decisions.22
New antibodies detect BRAF V600E mutations that facilitate melanoma growth. These markers help with diagnosis and treatment choices, as BRAF inhibitors work well with patients with these mutations.23
Integrating standard melanocyte markers with new molecular markers improves melanoma testing, diagnosis, assessment, and treatment selection.
Recent advances in melanocyte biology have identified promising biomarkers with research and clinical potential. These markers highlight melanocyte development, function, and pathogenesis.
ABCB5 (ATP Binding Cassette Subfamily B Member 5) is a membrane transporter protein expressed on melanoblasts and corresponding stem cells. ABCB5-positive cells can push out chemotherapy drugs (chemoresistance) and, in melanoma models, proliferate more quickly.24 Antibodies targeting ABCB5 help find melanocyte stem cells and target hard-to-treat melanoma subpopulations. Current clinical trials are evaluating ABCB5 as a therapeutic target in advanced melanoma.
AXL receptor tyrosine kinase serves as a marker for invasive and treatment-resistant melanomas. AXL expression increases when melanoma cells change from proliferative to invasive.25 IHC detection of AXL pinpoints aggressive melanomas and helps guide treatment choices, particularly for patients likely to benefit from AXL inhibitors currently in clinical development.
NFIB (Nuclear Factor I B) is a transcription factor that causes melanoma to spread. It upregulates genes that help cells migrate and mediate extracellular matrix interactions.26 Antibodies targeting NFIB could detect melanomas that are more likely to spread.
L-PGDS (Lipocalin-type Prostaglandin D Synthase) is a newly found melanocyte marker that works with standard markers. Researchers have identified L-PGDS expression in melanocytes in skin and hair follicles, suggesting it plays an important functional role that requires further study.27
These emerging markers are at the cutting edge of melanocyte research. While they need more testing before regular clinical use, they show how research keeps adding to our tools for finding and studying melanocytes.