Lucas Baumard, PhD | 12th May 2025
T helper cells are an important T cell subset of the adaptive immune system. They can be broadly categorized into Type1, Type 2 and Type 17 helper cells, recognized primarily by the cytokines they release (Table 1). Characterized by their CD4 expression (a pan T helper cell marker), T helper cells can activate, suppress, or induce differentiation in other immune cells mainly through the cocktail of cytokines they secrete. These subsets are specialized in their responses to specific pathogens.1
Naïve CD4+ T cells are able to differentiate into one of several different subsets (Figure 1) following activation by antigen-presenting cells such as dendritic cells.
Figure 1: Differentiation pathways of naïve CD4+ T cells (Th0). Transcription factors and release cytokines for each subtype are indicated. Top cytokines drive differentiation of each subtype. Type 1 immune response: defense against intracellular bacteria, protozoa and viruses; type 2: defense against helminths and venoms; type 3: defense against extracellular bacteria and fungi.
Along with the nature of the specific antigen and local cytokine signals, T cell receptor binding is one of the main drivers of differentiation that determines CD4+ T cell fate. Weak affinity antigen presentation via MHC has been shown to favor Th2 differentiation whilst strong antigen presentation favors Th1 differentiation.2 Differentiation drives the release of Th subset-dependent cytokines, which in turn drive a positive feedback loop of increased differentiation via transcription factor upregulation (Table 1).
| CD4+ Subset | Surface markers | Cytokines | Transcription factors |
|---|---|---|---|
| Th1 | CCR5, CXCR3 | IL-2, IL-12, IFNγ | T-bet, STAT1, STAT4, Runx 3, Eomes, Hlx |
| Th2 | CCR4, CRTh2 | IL-2, IL-4 | GATA3, STAT6, STAT5, STAT3, Gfi-1, c-Maf, IRF4 |
| Th9 | None specific | IL-4, TGF-β | IRF4 |
| Th17 | CCR4, CCR6 | IL-6, IL-17, IL-21, IL-23, TGF-β | RORγt, STAT3, RORα, Runx1, Batf, IRF4, AhR |
| Th22 | CCR4, CCR6, CCR10 | IL-13, IL-22, TNF-α, Granzyme B | AhR, RORγt, T-bet, Runx1, HIF-1α |
Table 1: Subsets of CD4+ T helper cells and their associated markers. Markers in bold indicate: classical surface markers, cytokines that drive self-differentiation, and master transcription factors. Edited from 3.
Because differentiation can be cytokine-driven, there is also measure of plasticity to T helper cells. Research has shown that T helper subsets are not only able to dynamically change their phenotype (including cytokine profile)4,5 but also express markers of other lineages.6,7 This adds an element of complexity to their study and characterization – particularly in inflammatory disease.
T helper cells can be characterized by the surface markers they express and the cytokines they release (Table 1). T helper cells can therefore be examined with a variety of experimental techniques. Surface marker expression can be detected by flow cytometry, IHC or western blot. Cytokine expression can be measured by ELISA, ELISPOT or intracellular flow cytometry. Because of the plastic nature of T cells, especially when it comes to surface marker expression, a combination of these markers and techniques might be best to accurately characterize these populations.
Th1 cells are a CD4+ T cell subset characterized by the release of the cytokines IFN-γ, TNF-α and IL-28 and the expression of the chemokine receptors CXCR3 and CCR5. Broadly they are characterized as pro-inflammatory cells.
Th1 cells can recognize antigens presented on MHCI/II by other immune cells such as dendritic cells.9 These cells are mostly responsible for combating intracellular bacteria and viruses (tuberculosis, Listeria etc.) that have infected cells. They also activate other cells of the immune system such as macrophages and B cells via the CD40 ligand and the release of cytokines.10
Th1 Cell Surface Markers
CXCR3 is a canonically described Th1 cell marker. The chemokine receptor is expressed on Th1 CD4+ cells, CD8+ T cells, natural killer (NK) cells and subsets of dendritic and B cells.11,12 CXCR3 is upregulated on naïve cell activation and binds the chemokines CXCL9, CXCL10 and CXCL11,13 which direct T cell migration. Expression of CXCR3 is increased in inflamed tissues14 and associates strongly with IFN-γ releasing cells.15 Therefore, levels of CXCR3 will be lower in inactive T cells in a non-inflammatory environment which reduces their utility as a Th1 cell marker outside of these conditions.
Though classically used as a Th1 cell marker, CXCR3 has been shown to be expressed on Th2 cells16,17 making this marker poor for Th1 cell characterization.
CCR5 (or CD195) is highly expressed in Th1 cells but it is less commonly used as a Th1 marker compared to CXCR3, being more recently discovered. Unlike CXCR3 however, Th2 cells are almost completely CCR5-null,16 making it a potentially stronger marker within CD4+ populations. CCR5 is also expressed on macrophages, dendritic cells, T memory cells as well as various other cells such as endothelial, epithelial cells and neurons, microglia.18
CCR5 is a chemokine receptor that directs cells to sites of inflammation, but unlike CXCR3 is not necessary for Th1 cell development.19 Ligands for the receptor include MIP1-α, -β, monocyte chemotactic protein two and RANTES.20-22
Figure 2: ICC/IF of HeLa cells stained with Anti-CD195 Antibody [12D1] (A248230).
Figure 3: Flow cytometry of human peripheral blood stained with Anti-CCR5 Antibody [T21/8] (APC) (A86073).
Th1 Cytokines
As well as their surface marker expression, Th1 cells can be identified by the specific cytokines they release to mediate immune responses.
Interferon gamma (IFN-γ) is one of the most important T helper cytokines. IFN-γ is a type II interferon cytokine released by Th1 and NK cells in response to IL-12, IL-18, pattern recognition receptor activation or microbial antigens.23 The receptor, IFN-γR, is expressed on almost all cell types24 - an indication of the importance of the cytokine. IFN-γR stimulation drives the transcription of a wide variety of genes that encode other immune responses e.g. cytokine release, antigen-presenting proteins.25 IFN-γ activates macrophages, other T helper cells (including Th1 differentiation) and B cells and downregulates Th2, Th17 responses.23,24
IL-2 is a general T cell growth factor which promotes the differentiation and proliferation of Th2 cells as well as CD8+ cytotoxic and memory T cells.26-28 IL-2 is released from activated Th1 cells and, to a lesser extent, CD8+ T cells, dendritic cells and B cells.29 IL-2 is generally anti-inflammatory, skewing the Th1: Th2 ratio towards Th2 cells (mostly anti-inflammatory) and increased expression of FOXP3 (drives Treg differentiation) in naïve CD4+ cells.30
Tumor necrosis factor alpha (TNF-α) is another cytokine released by Th1 (and Th17) cells that has an inhibitory and proliferative effect on Th1 and Th17 cells respectively.31 TNF-α mediates its effect through Tumor necrosis factor receptor 1 and 2 with TNFR1 being expressed on almost all cells whilst TNFR2 is rarely expressed outside the brain.32 TNF-α has been show to activate inflammatory responses, namely the apoptosis and necroptosis pathways in both diseased as well as excessively inflammatory cells.33
Th2 cells are a CD4+ T cell subset that drive type 2 inflammatory or anti-inflammatory responses through the recruitment of mast cells, basophils and eosinophils,34 and release of IL-4,35 IL-5,36 IL-25 and IL-33.37 Th2 cells are best known for their role in combatting early helminth infections but are also involved in autoimmune and allergic responses (particularly asthma).
Th2 Cell Surface Markers
Though there are canonical Th2 cell surface markers, they share some of the same issues in terms of plasticity as discussed for Th1 markers. For instance, some T cells have been shown to co-express CXCR3 as well as the Th2 markers CCR438 and CRTh2.39 In addition, Th2 cell markers are particularly promiscuous, being expressed on various other T cell subsets. Therefore, it is likely necessary to combine data from the expression of multiple surface receptors as well as cytokine data to accurately characterize Th2 cells.
CCR4 is a chemokine receptor expressed on IL-4, IL-5, and IL-13 producing Th2 cells40,41 as well as Tregs,42 Th1743 and Th22 cells.44 Activation of the receptor by the ligands CCL17 and -22 lead to its internalization.45 The receptor is upregulated in asthmatic airways40 and certain lymphomas.42
CRTh2 (or prostaglandin DP2 receptor) is a relatively novel receptor for prostaglandin DP2 expressed on Th2 cells but also CD8 cells, eosinophils, and basophils.46 Much like CCR4, the expression and phenotype is complex, but it has been shown to be a more accurate Th2 marker than CCR4.46 In one study, CRTh2+ cells produced the Th2 cytokines IL-4, -5 and -13, but not the Th1 cytokine IFN-γ. On the other hand, CCR4+ cells express moderate amounts of IFN-γ and show reduced IL-4 and IL-13 expression.46
Figure 4: IHC of human bladder carcinoma tissue stained with Recombinant Anti-GATA3 Antibody [GATA3/4550R] (A277989).
Figure 5: Flow cytometry of Expi293 cells transfected with human CCR4 (blue) or irrelevant protein (red) stained with Recombinant Anti-CCR4 Antibody [DMC297] - BSA and Azide free (A318770).
Th2 Cytokines
Like Th1 cells, Th2 cells express a specific combination of cytokines to help mediate type 2 immune responses and activate other effector cells such as B cells and mast cells (Figure 6).
Figure 6: Activation of Th2 cells by antigen presentation from dendritic cells (DCs) and their downstream effects.
IL-4 is a cytokine important for the development of Th2 cells: IL-4 gene expression is necessary for the development of naïve CD4+ T cells48 but is produced in low amounts in recently differentiated populations49 until activation when release increases.50 IL-4 is also released by eosinophils,51 mast cells and basophils52 in response to parasitic infection, as well as by T follicular helper cells. IL-4 released from the latter has been shown to induce IgE and IgG1 antibody responses whereas Th2-derived IL-4 does not.53 IL-4 is also important in wound healing, nerve regeneration and allergic disease.54
IL-13 works in a similar manner to IL-4, driving type 2 inflammatory responses and is an important cytokine in allergic disease. Th2 cells generally co-express IL-4 and IL-13.55 During helminth infections, however, they work differently, with IL-4 being more important in IgE production and mast cell activation, whilst IL-13 acts to increase mucus production, goblet cell hyperplasia and the physical expulsion of worms.56-58 IL-13 alone has been shown able to drive airway hyperreactivity in asthma.59
IL-5 is released from Th2 (not Th1)60 cells, mast cells and eosinophils.61 IL-5 specifically activates eosinophils, basophils and B cells as these cells preferentially express IL-5R.62,63 IL-5 is required for proliferation and Ig production in B cells.64
IL-25 is a cytokine that drives type-2 immune responses65 and is frequently studied in allergy and autoimmune diseases. IL-25 is released by Th2 cells in response to helminth infection and acts in an autocrine manner to induce the release of IL-4, IL-5 and IL-13.66 IL-25 signaling also regulates T cell and proliferation via L-arginine metabolism pathways.67
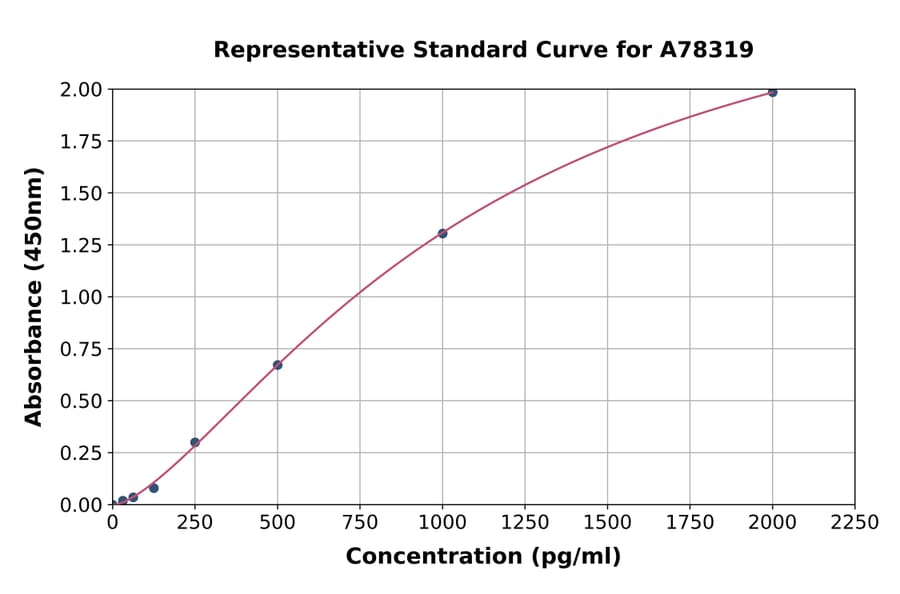
Figure 7: Representative standard curve for Human IL-4 ELISA Kit (A78319).
Figure 8: Flow cytometry of viable human lymphocytes stained with Anti-IL-4 Antibody [IL4-3] (PF488P) (A269813) after stimulation with PMA/ionomycin (right) compared with no stimulation (left).
Th17 cells are a further subset of CD4+ T cells that are characterized by the cytokine IL-17. Th17 cells are pro-inflammatory and are protective against extracellular bacteria and fungal infections, but pathogenic in various autoimmune diseases.68 Th17 differentiation is driven by TGF-β and IL-21.69
Th17 Cell Surface Markers
Th17 cells are typically classified as CD4+CCR4+CCR6+. Th17 cells share CCR4 with Th2 (and Th1 to an extent) cells, but CCR6 is more specific to Th17 cells, as well as being expressed on Treg cells, dendritic cells and B cells.70 Pro-inflammatory Th17 cells and anti-inflammatory Treg cells both express CCR6, though Tregs do not typically release IL-17 or other Th17 cytokines.71 The expression of the CCR6 ligand, CCL20, on Th17 cells, drives the migration of both Tregs and Th17s to inflammatory sites,72 the balance of which determines inflammatory response. This balance is skewed towards Th17s in disease. The CCR6-CCL20 axis is therefore of particular clinical interest, with inhibitors to one side of the axis trialed in diseases from cancer to arthritis and diabetes.72
Th17 Cytokines
Th17 cells release IL-17 (in its two isoforms IL-17A and IL-17F), IL-21 and IL-22.
IL-17A and IL-17F are the only members of the IL-17 family released by T cells. These cytokines induce the expression of the chemokines CXCL1, CXCL2, CXCL5, and CXCL8, and the granulpoietic factors G-CSF and stem cell factor in epithelial cells.73,74 IL-17A also increases mucin production in bronchial airways.75 The cytokines therefore play an important role in inflammation and responses to pulmonary infections. In pathology, they are associated with psioriasis,76 arthritis and driving destruction of cartilage and bone.77
Figure 9: An ELISpot analysis of cells secreting IL-17A and Bovine PBMC incubated overnight in the presence (40,000 cells per well) or absence (200,000 cells per well) of PHA. Staining performed with Anti-IL-17A Antibody [MT49A7] (A269864).
IL-21 is released by natural killer T cells, Th17 cells, and other CD4+ subsets.78,79 IL-21 has been shown to drive Th17 development in an autocrine, positive feedback loop and promote Th1 responses (namely IFN-γ production).80,81 IL-21 favors Th17 generation over Treg cells.78 IL-21 is also important in IgG1 and IgE-dependent B cell activity and NK development.82,83 IL-21 is associated pathologically with Systemic lupus erythematosus (SLE), Type 1 diabetes, and inflammatory bowel disease (IBD).78
IL-22 is a member of the IL-10 family of cytokines and is released from the CD4+ Th17, Th1 and Th2 cells (ordered of highest to lowest).84 It is released in response to IL-6 and IL-23 and is important in early host responses to bacteria through regulation of antimicrobial proteins.85,86 The cytokine is also upregulated in Crohn’s disease, ulcerative colitis and IBD.87-89
Diagrams created with BioRender.com.

![IHC - Anti-CD195 Antibody [12D1] (A248230)](https://cdn.antibodies.com/image/catalog/248/A248230_5.jpg?profile=product_image)
![Flow cytometry - Anti-CCR5 Antibody [T21/8] (APC) (A86073)](https://cdn.antibodies.com/image/catalog/86/A86073_413.jpg?profile=product_image)
![IHC - Recombinant Anti-GATA3 Antibody [GATA3/4550R] (A277989)](https://cdn.antibodies.com/image/catalog/277/A277989_1.jpg?profile=product_image)
![Flow cytometry - Recombinant Anti-CCR4 Antibody [DMC297] - BSA and Azide free (A318770)](https://cdn.antibodies.com/image/catalog/318/A318770_1.png?profile=product_image)

![Anti-IL-4 Antibody [IL4-3] (PF488P) (A269813)](https://cdn.antibodies.com/image/catalog/269/A269813_2.jpg?profile=product_image)